Biomarker testing has transformed cancer care over the past decade, opening the door to targeted therapies that can improve outcomes for patients with advanced disease. Yet despite strong guideline recommendations, many patients with metastatic non-small cell lung cancer (NSCLC) may not receive timely testing.
In this Truveta experts spotlight, Amy Sullivan, MS, Partner-Facing Research Analyst at Truveta, brings her background in biostatistics, genomics, and real-world evidence to examine biomarker testing trends in metastatic NSCLC. Her analysis of Truveta Data highlights both progress and persistent gaps in ensuring timely testing for all patients.
Why it matters: The role of biomarker testing in NSCLC
Patients with metastatic NSCLC can benefit from therapies tailored to the presence of a specific mutation or fusion. Diagnostic and pharmaceutical companies have invested heavily in these tests but often have a limited understanding of the real-world usage and changes in trends. Timely biomarker testing is essential to ensure patients receive the most appropriate treatment.
Methods
Using Truveta Data, Amy examined patients newly diagnosed with metastatic NSCLC between January 1, 2018, and December 31, 2024. A total of 9,824 patients were included in the analysis. Both squamous and non-squamous NSCLC patients were grouped together, with no additional inclusion or exclusion criteria applied.
The analysis measured whether patients had a biomarker test within 90 days before or after their first metastatic NSCLC diagnosis and calculated the annual proportion of patients receiving such testing.
Results
Across the study period, 3,739 patients (38%) received biomarker testing within 90 days of their metastatic NSCLC diagnosis. Early in the observation period, testing was relatively uncommon: in 2018, just 27.9% of patients received a test. Rates climbed steadily through 2021, peaking at 40.8%. However, since 2021, this progress has leveled off, with proportions stabilizing between 38% and 42% through 2024.
Among patients who received biomarker testing, 49% were male, 45% female, and 6% recorded as unknown—a distribution that mirrors the overall demographics of metastatic NSCLC in the U.S. population.
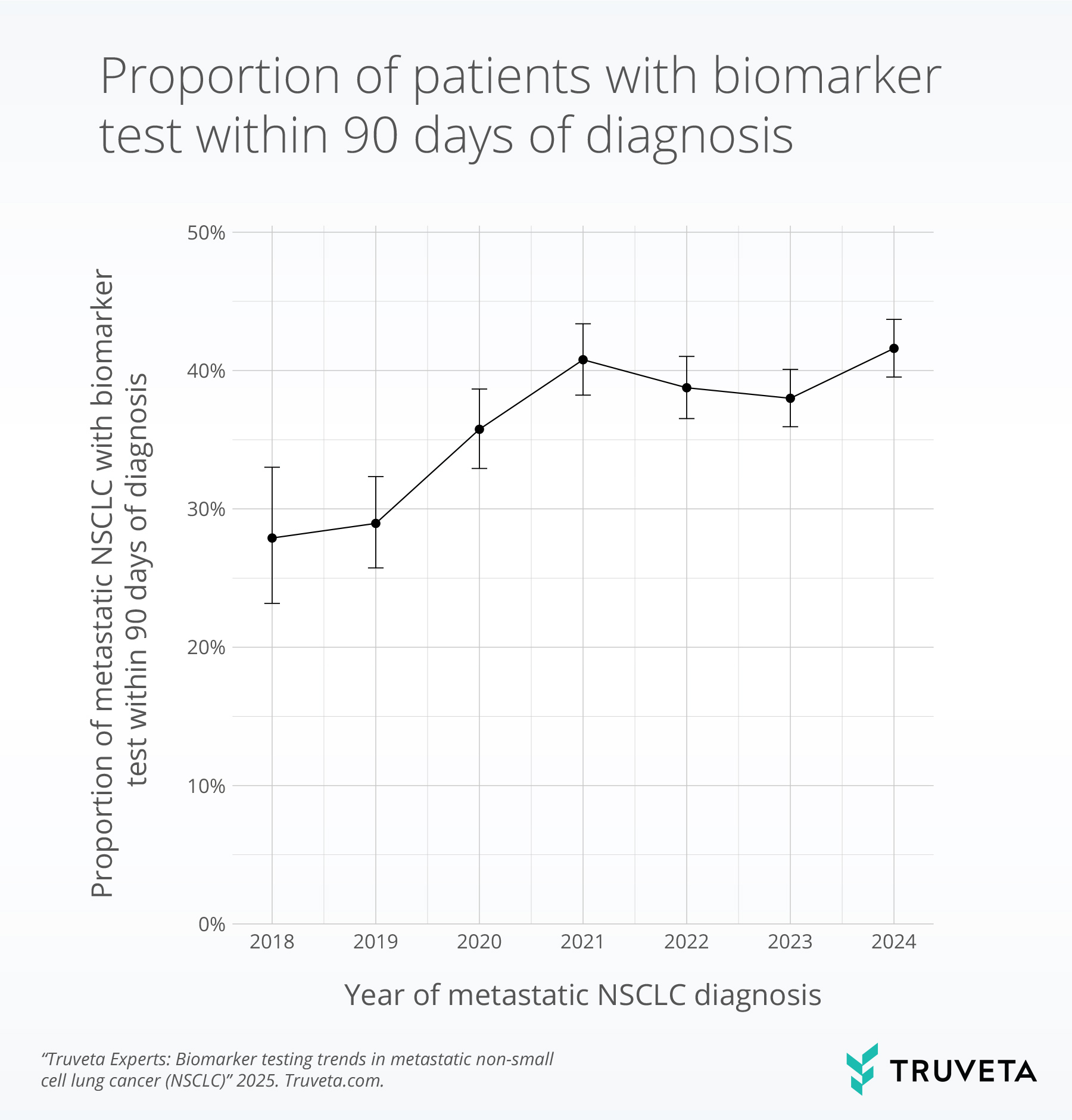
Figure 1. Proportion of patients with metastatic NSCLC who received biomarker testing within 90 days of diagnosis, 2018–2024. Error bars represent 95% confidence intervals.
Discussion
These results show that while biomarker testing rates have improved over time, fewer than half of patients with metastatic NSCLC are tested within the recommended 90-day window. This gap suggests that many patients may miss the opportunity for targeted therapies that could significantly impact outcomes.
The plateau in testing rates since 2021 underscores both progress and persistent challenges. Despite biomarker testing being standard of care, real-world uptake appears to have reached a ceiling, highlighting potential barriers related to provider practices, testing infrastructure, payer coverage, or patient access.
The narrower confidence intervals in later years indicate greater precision of those estimates, which is consistent with larger effective samples sizes and/or lower variability in those periods. Ensuring broader adoption of biomarker testing remains a critical opportunity to expand access to life-saving targeted therapies.
Future research
Future research directions could include evaluating patient outcomes in relation to biomarker testing patterns, conducting a more detailed assessment of the types of tests employed (e.g., immunohistochemistry versus molecular profiling assays), and examining test results in the context of other routinely collected clinical measures, such as laboratory values and vital signs—all available within Truveta Data.
About Truveta experts
The Truveta experts series highlights insights from researchers, clinicians, and data scientists working across Truveta. Each post highlights how real-world data can unlock new understanding—from disease patterns to treatment complexity to population health trends.
Explore more in this series:
Treatment trends for trigeminal neuralgia
Fireworks during the fourth of July
Real-world trends in Rituxan biosimilar adoption
Sudden cardiac arrest and arrhythmias among patients with chronic kidney disease
See how Truveta Data can accelerate your next study—Request a customized demo today

