Saving Lives with Data
Where yesterday's care becomes today's intelligence
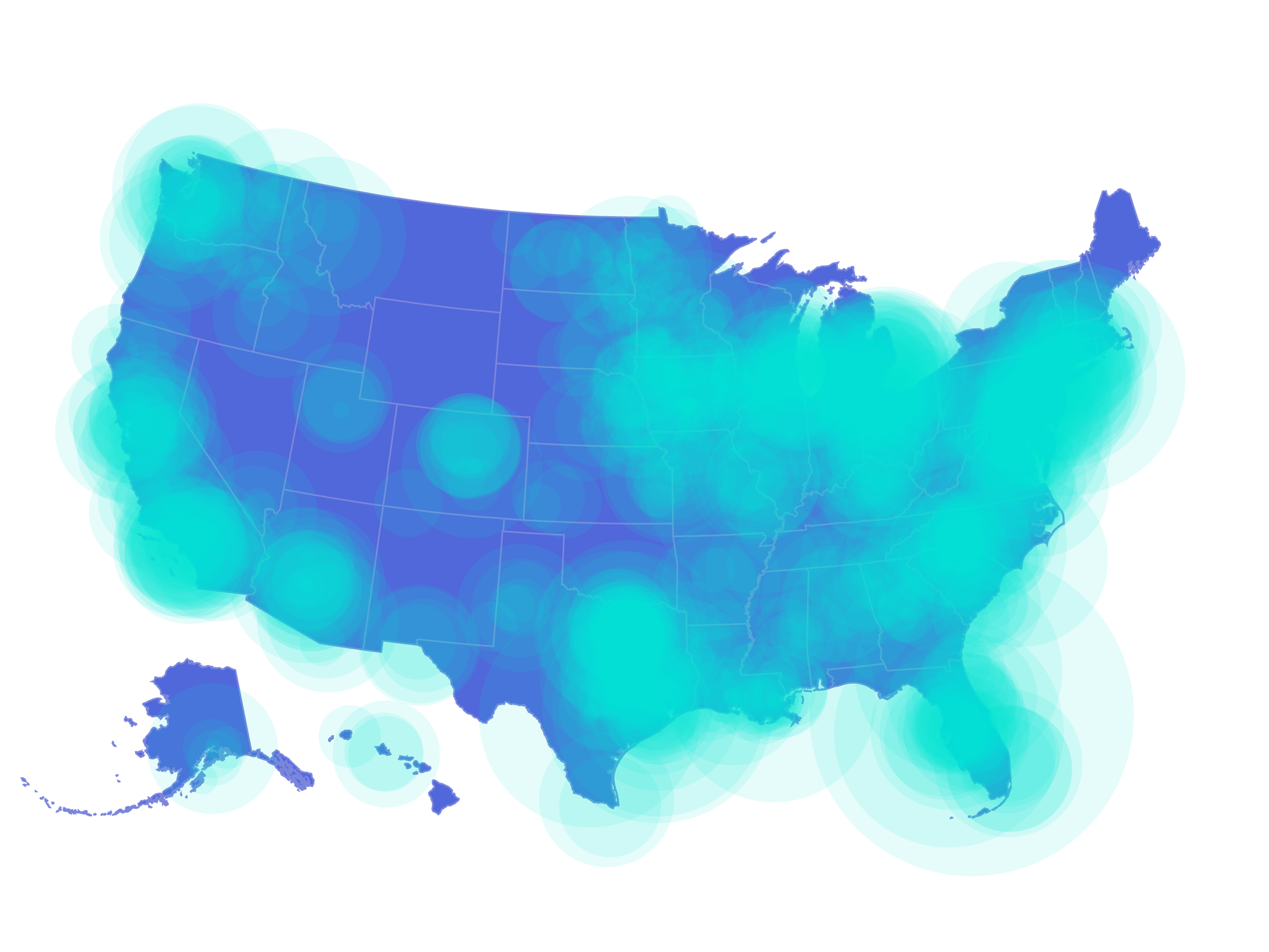
Truveta Data
The most complete, real-time view of US healthcare
130M+
Patients and growing
Updated daily
Longitudinally linked:

EHR

Closed claims

Devices

Images

SDOH

Mother-child

Multiomics

Mortality
Truveta Intelligence
Answers in minutes, not months
Truveta Evidence
Confidence through rigorous analytics and expertise
Trusted research environment

Immediately access Truveta Data and execute full research studies

Dynamically refine cohorts with reusable, clinically precise definitions

Produce audit-ready evidence aligned to regulatory standards
Truveta Evidence Services
Design rigorous studies alongside experts and deliver transparent, audit-ready evidence

Extend capacity with trusted experts

Accelerate timelines from protocol to submission

Maximize value from real-world data

De-risk high-impact regulatory and post-market studies
350+
Publications
250+
Conference presentations
150K+
Citations
100+
Regulatory projects across US and global agencies
Power every stage of care
Advance every decision with real-time intelligence and regulatory-grade evidence
Discovery
Discover new targets and train AI
Therapy adoption
Track patient access and industry trends in real time
Safety
Uncover safety signals faster from real-time data trusted by regulators
Outcomes research
Update evidence strategies in real-time across precise populations
Clinical trials
Simulate trials and identify eligible patients with unprecedented precision
Healthcare optimization
Benchmark care quality and enhance operations
Built with US health systems






























Pharmaceutical

Medical device

Healthcare

Public health

Academics
Ready for the entire healthcare ecosystem
Ready for the entire healthcare ecosystem

Pharmaceutical

Medical device

Public health

Healthcare

Academics
Trusted by leading life science, public health, and healthcare organizations
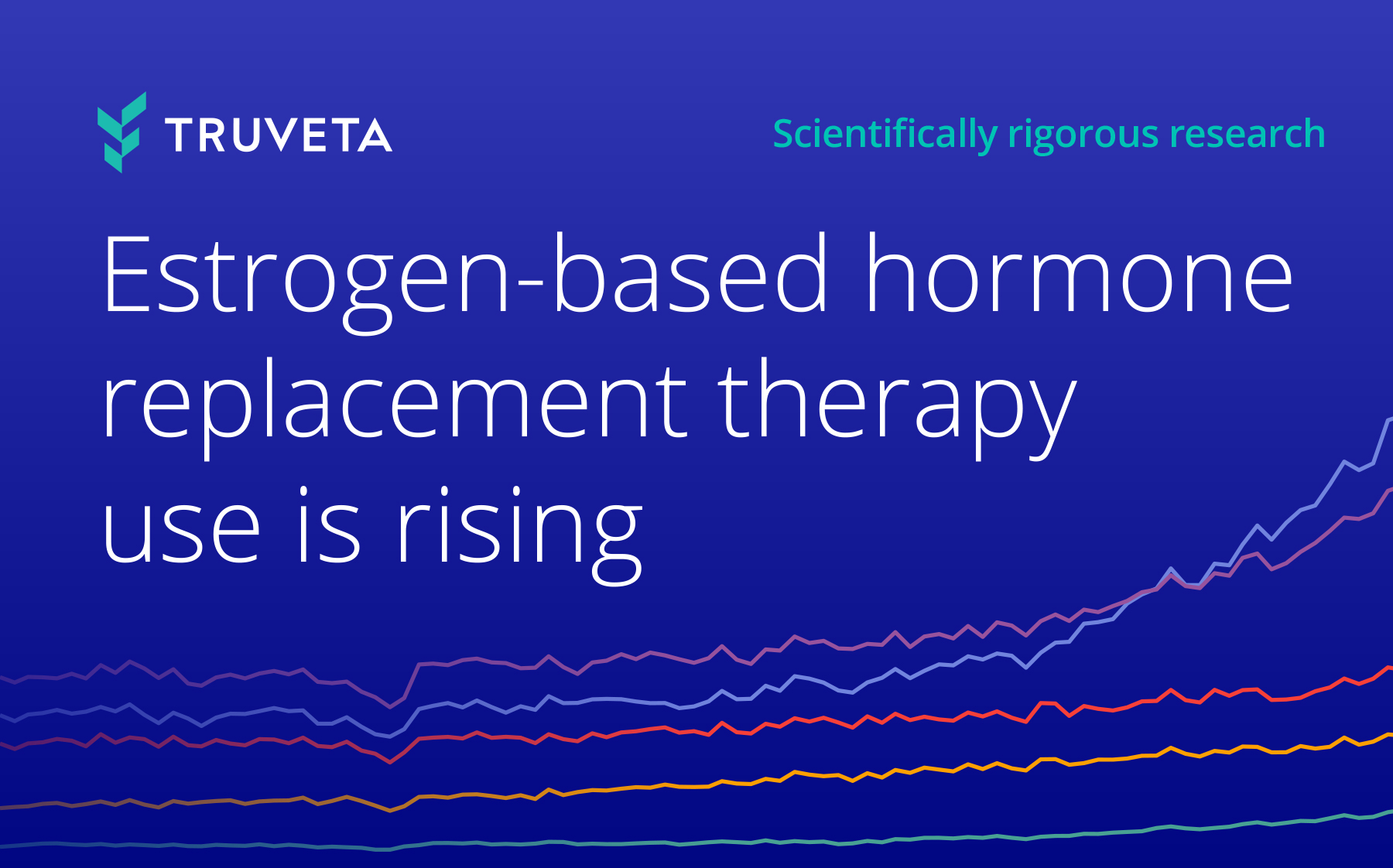
Featured research
Featured in