Clinical trials rarely stall because of science alone. More often, they slow due to recruitment challenges—particularly in rare and clinically complex conditions.
When that happens, knowing that eligible patients exist is not enough. Sponsors need to locate eligible patients quickly, and work to activate sites, in order to deliver potentially life-saving treatment faster.
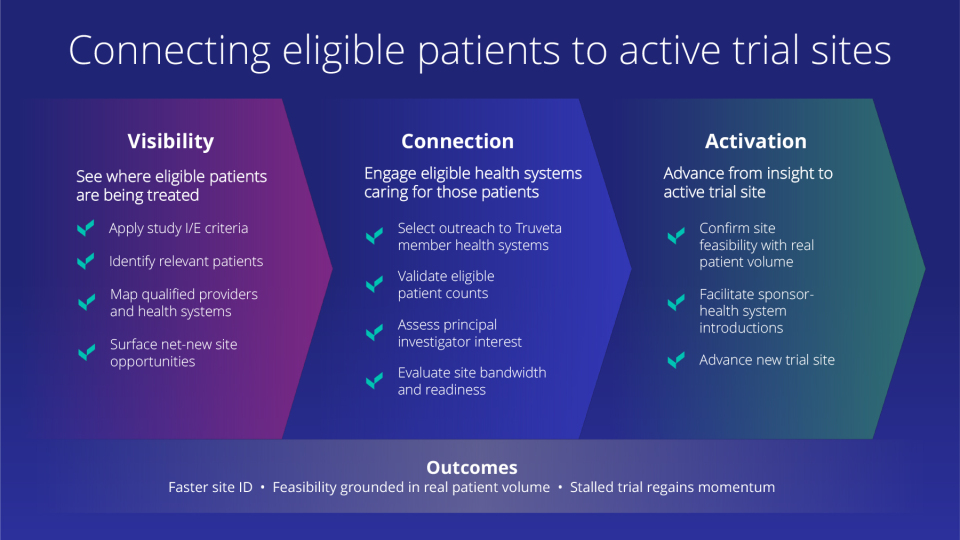
Truveta’s clinical trials approach accelerates this identification and enrollment process. By pairing representative, daily updated real-world data with active health system partnerships, sponsors can move from identifying patients to engaging the sites that care for them.
A recent Phase 2 study in a rare hematologic disease illustrates how that shift can help a stalled trial regain momentum.
Finding patients where they are treated
One such sponsor was looking to enroll trial patients with a rare blood disorder characterized by progressive anemia and transfusion dependence. The study initially enrolled approximately 100 patients across 15 active sites. However, recruitment of individuals with high-transfusion dependence—a group critical for evaluating therapeutic impact—proved difficult and slower than expected.
To address this gap, the study sponsor partnered with Truveta to apply the study’s inclusion and exclusion criteria to longitudinal electronic health record data. Using daily updated EHR data enriched with laboratory values and transfusion history, Truveta evaluated where eligible patients were currently being treated and which providers were managing them.
The analysis identified health systems with meaningful concentrations of eligible patients, including Baylor Scott & White Health.
The missing link: Direct health system engagement
Identifying a high-volume of eligible patients was step one. Because Truveta is uniquely built with and owned by US health systems, the analysis transitioned directly into a sponsor-site engagement. Study leaders reviewed patient volume, confirmed investigator interest, and assessed site bandwidth. This work ultimately advanced Baylor Scott & White as a net-new clinical trial site for the sponsor.
In parallel, Truveta supported development of a real-world external control arm to characterize anemia progression and transfusion dependence in comparable patients. These analyses will inform future Phase 3 trial design.
In this example, Truveta helped:
- Identify new trial sites with demonstrated access to eligible patients
- Enable faster, feasibility-driven site selection that advanced into contracting
- Build an external control arm to inform future Phase 3 trial design
A new solution for stalled clinical trials
Enrollment challenges in clinical trials are frequently problems of visibility and alignment. With access to the most representative, complete, and timely patient journey data, sponsors can see where eligible patients are being treated—and now engage those sites directly.
As inclusion criteria grow more specific and therapies target narrower populations, the ability to connect real-world data with the health systems delivering care becomes increasingly important. With these new capabilities, sponsors can design smarter studies, enroll patients faster, and generate the robust evidence needed to bring safer, more effective therapies to patients—more efficiently than ever before.

