Authors: Karen G Farrar ⊕University of Southern California, Los Angeles, CA, Duy Do ⊕Truveta, Inc, Bellevue, WA, Nina B Masters ⊕Truveta, Inc, Bellevue, WA, Brianna M Cartwright ⊕Truveta, Inc, Bellevue, WA
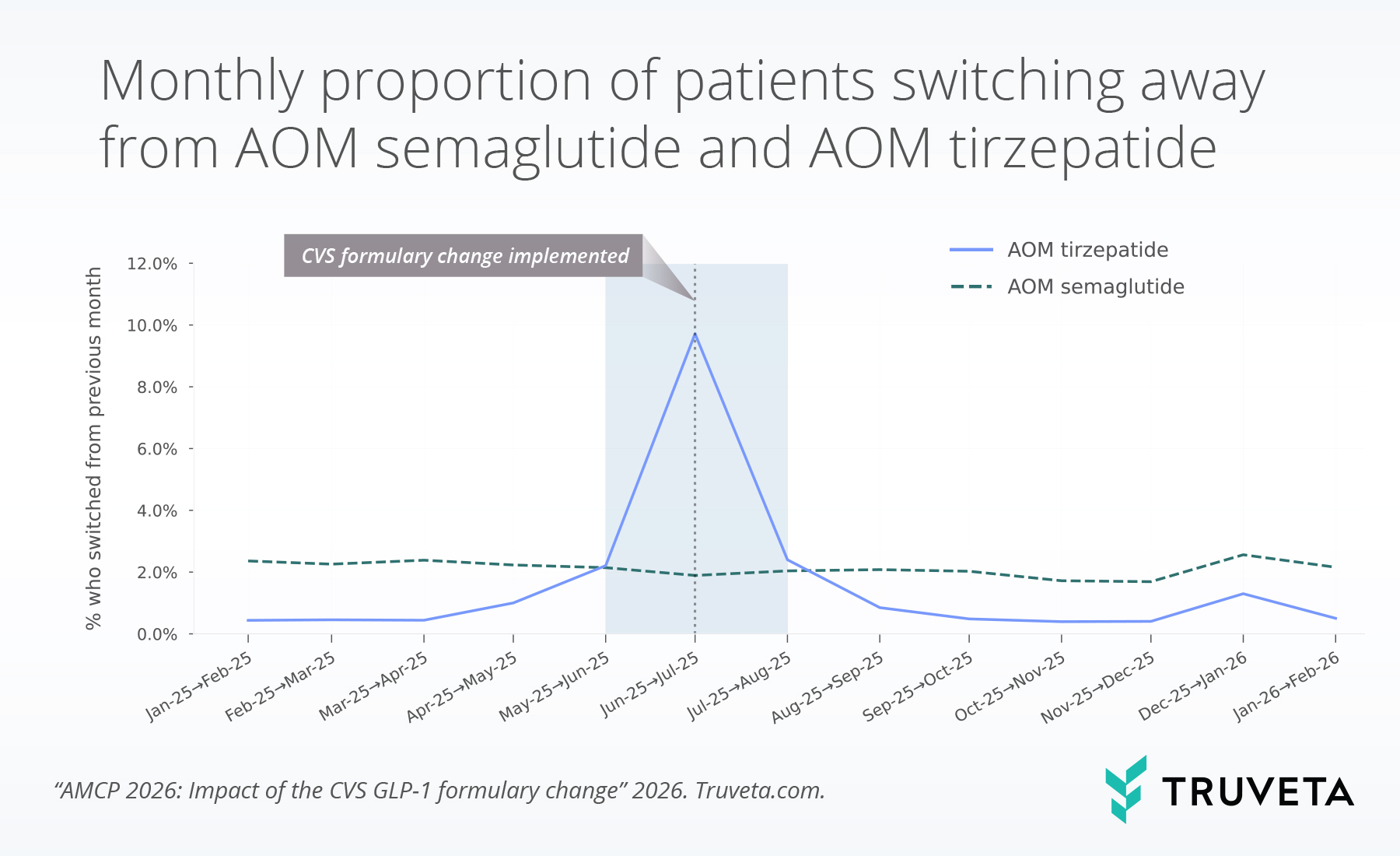
- After CVS Caremark made anti-obesity semaglutide the preferred formulary option in July 2025, switching from anti-obesity tirzepatide surged from 0.6% to 9.7%, representing more than a 16-fold rise.
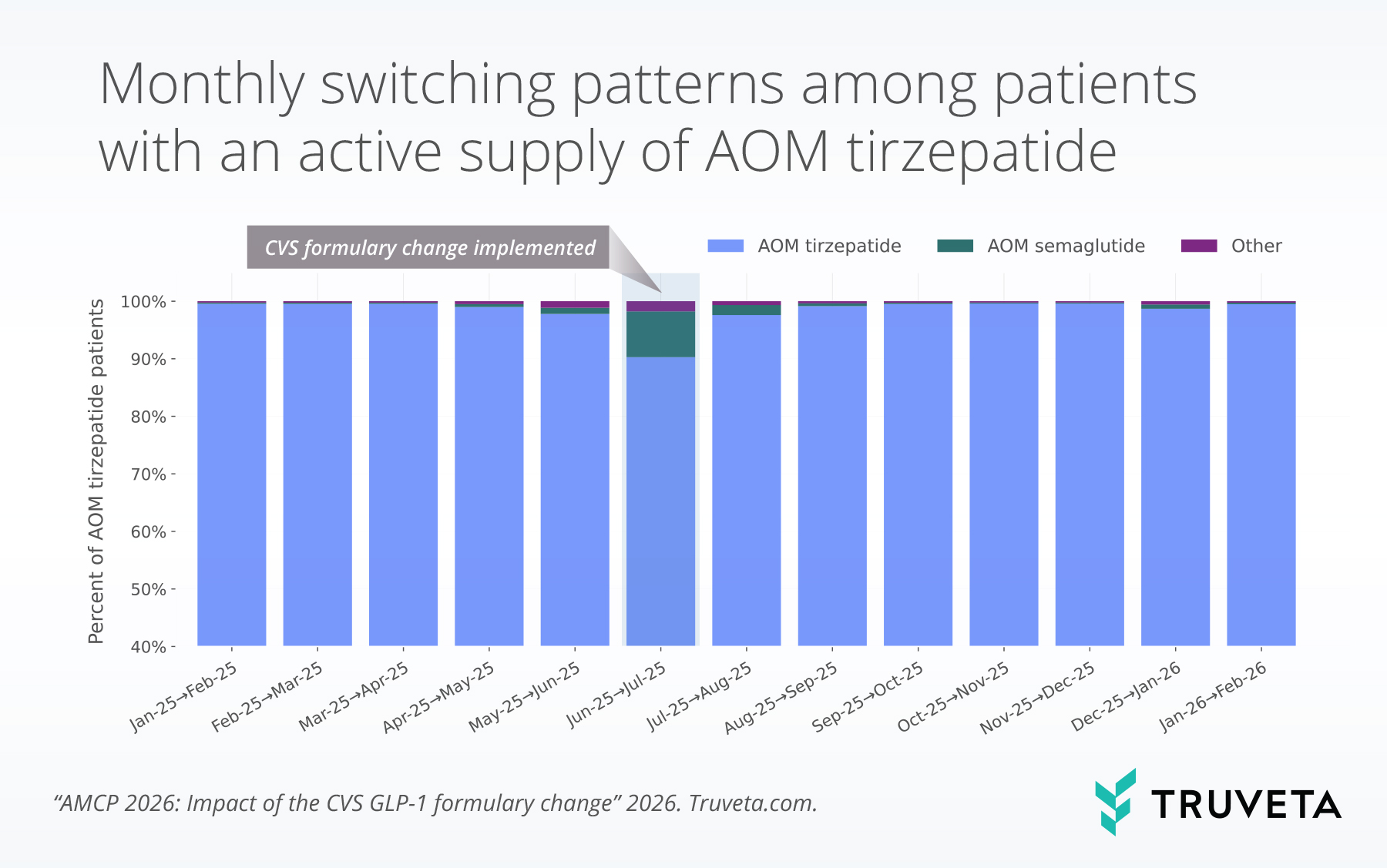
- Of the patients who switched medications, 8 in 10 switched to AOM semaglutide.
- Following this spike, switching rates declined after the spike but stayed elevated (~1.6%) through September before returning to baseline in October 2025.
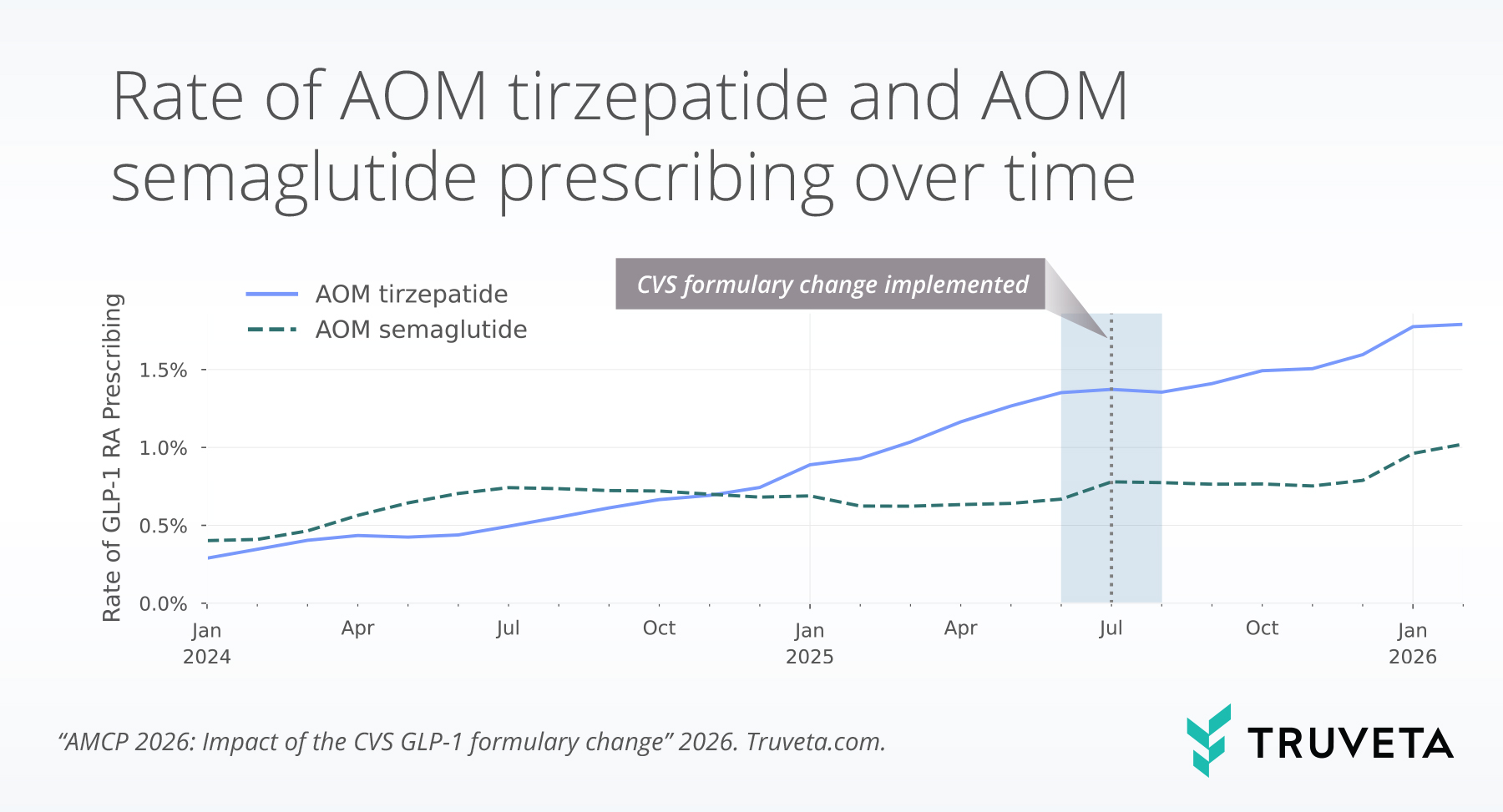
- Prescribing trends mirrored this pattern: AOM semaglutide saw its largest monthly increase in July, stabilized from September–November, then rose modestly in November–December and accelerated through February 2026.
This report is an extension of our poster presented at AMCP, titled Impact of the CVS GLP-1 formulary change.
Glucagon-like peptide-1 receptor agonist based medications (GLP-1s) have transformed the treatment landscape for type 2 diabetes and obesity over the past decade (1, 2). These medications have demonstrated substantial benefits in glycemic control, weight reduction, and cardiovascular outcomes, contributing to their rapid uptake in clinical practice (1, 3). Our recent analysis shows that GLP-1s now account for more than 7% of all prescriptions in the US, reflecting both expanding indications and increasing demand (4).
Despite their clinical effectiveness and the volume they represent in the market, real-world use of GLP-1s is heavily influenced by non-clinical factors such as drug availability, insurance coverage, and formulary decisions (5, 6). These factors can shape not only which medications are prescribed, but also whether patients remain on therapy or transition to alternatives.
In May 2025, CVS Caremark announced that anti-obesity (AOM) tirzepatide (sold as Zepbound) would be removed from its preferred formulary effective July 1, 2025, with AOM semaglutide (sold as Wegovy) designated as the preferred alternative. Importantly, this decision applied only to the obesity indication (7, 8). Tirzepatide remained available for patients with type 2 diabetes under its anti-diabetic (ADM) formulation (sold as Mounjaro).
We previously examined GLP-1 dispensing and prescribing patterns immediately following the July 2025 CVS formulary change and found that switching from AOM tirzepatide increased more than 16-fold in July, with most patients transitioning to AOM semaglutide. At the same time, prescribing trends shifted, with AOM semaglutide experiencing its largest monthly increase while AOM tirzepatide growth slowed and initiations declined. Building on this prior work, we extended our prior analysis to look at more recent patterns, as time since the formulary change has passed.
Methods
We conducted two complementary analyses using subsets of Truveta Data.
Switching population
We identified patients aged 18 years or older with overweight or obesity (BMI >27) and at least one GLP-1 medication dispense between January 2025 and February 2026. Medications included semaglutide (AOM and ADM, oral and injectable), liraglutide (AOM and ADM), exenatide (ADM), lixisenatide (ADM), tirzepatide (AOM and ADM), and dulaglutide (ADM). Medications were classified by brand name to determine indication (AOM vs. ADM). Dispense data included prescriptions filled both within and outside Truveta health systems.
Active medication supply was constructed using dispense dates and days’ supply. For prescription fills exceeding 30 days, we assumed supply extended across subsequent months (e.g., a 60-day fill was counted as two months of supply), referred to as “active supply.” Patients were required to have at least two consecutive months of active supply. Switching was defined as a change to a different GLP-1 between consecutive months (e.g., January to February 2025).
We evaluated monthly switching patterns across consecutive months using two complementary approaches. First, among patients with an active supply of AOM tirzepatide or AOM semaglutide, we calculated the percentage who switched to a different GLP-1 in the subsequent month. Second, among patients with an active supply of AOM tirzepatide, we evaluated the distribution of GLP-1 use in the following month.
Prescribing population
We identified patients aged 18 years or older who were prescribed a GLP-1 between January 2024 and February 2026. Using prescription data, we evaluated both overall prescribing rates across all GLP-1s, with a focus on AOM tirzepatide and AOM semaglutide.
Results
Population characteristics
The switching population included 854,054 patients. Approximately half were aged 45–64 years (50.8%), 62.8% were female, and most identified as White (69.8%) and non-Hispanic or Latino (82.5%). Nearly half (48.0%) had type 2 diabetes.
The prescribing population included 2,227,978 patients, representing 9,376,370 GLP-1 prescriptions between January 2024 and February 2026.
Switching patterns following formulary change
Switching from AOM tirzepatide remained rare prior to May 2025, averaging 0.6% of patients who switched to a different GLP-1 in the following month. However, between June and July 2025, switching increased sharply to 9.7%, representing more than a 16-fold rise. Following this spike, switching rates declined but remained elevated through September 2025 at 1.6% on average before returning to an average baseline level of 0.6% between October 2025 and February 2026.
In contrast, switching among patients with an active supply of AOM semaglutide declined slightly from June to July 2025 (1.9%) compared to an average switching rate of 2.3% from January to April.
Among patients with an active supply of AOM tirzepatide, we observed a marked shift between June and July 2025. While most patients remained on AOM tirzepatide, 8.0% switched to AOM semaglutide during this period. Among those who switched therapies, more than 8 out of 10 (81.8%) patients switched to AOM semaglutide.
Prescribing trends over time
Prescribing patterns showed parallel shifts during the same period.
From January to June 2025, AOM tirzepatide prescribing increased steadily (+0.09 percentage points per month). However, growth slowed following the formulary change, increasing by just 0.02 percentage points from June to July and declining from July to August (−0.02 points). Prescribing then rebounded, with modest growth from September through November (+0.05 percentage points per month), followed by a more pronounced increase from December 2025 through February 2026 (+0.10 percentage points per month).
In contrast, AOM semaglutide prescribing remained relatively stable from January to June 2025, with minimal change over this period. From June to July, prescribing increased sharply (+0.11 percentage points), representing the largest single-month increase observed during the study period. Following this spike, prescribing was relatively stable from September through November (−0.01 percentage points per month), before increasing again from November to December (+0.04 points) and accelerating from December 2025 through February 2026 (+0.12 percentage points per month).
Discussion
This study provides a comprehensive view of how a major formulary change influenced both treatment switching patterns and overall prescribing rates for both AOM tirzepatide and AOM semaglutide.
Following the July 2025 CVS Caremark formulary change, we observed an immediate increase in switching from AOM tirzepatide, while patients with an active supply of AOM semaglutide showed stable or even slightly reduced switching during the same period, consistent with its designation as the preferred option. Among patients who switched from AOM tirzepatide to a different GLP-1, the majority transitioned to AOM semaglutide, aligning with its status as the CVS Caremark preferred therapy.
Prescribing patterns showed a parallel shift. AOM semaglutide prescribing increased sharply following the formulary change, while growth in AOM tirzepatide slowed and briefly declined. In the months immediately following, AOM tirzepatide prescribing rebounded, while AOM semaglutide prescribing returned to relatively stable levels after the initial increase. An additional increase in AOM semaglutide prescribing was seen in February, which could reflect uptake of oral semaglutide. We have a prior analysis demonstrating rapid uptake of oral semaglutide among patients new to taking GLP-1 medications within the first six weeks. These patterns may reflect shifts in prescribing behavior following updated coverage.
Importantly, these changes were short-lived. Rates of patients switching from AOM tirzepatide declined in the months following July 2025 and returned to near baseline levels by October, and growth in AOM tirzepatide prescribing rebounded after the initial decline. Together, these findings suggest a rapid but short-lived shift in switching and prescribing patterns following the formulary change.
This formulary-driven shift in obesity treatment patterns has important implications for patients. For some patients, maintaining coverage may require switching therapies, while others may face higher out-of-pocket costs to remain on their current medication. Tirzepatide and semaglutide differ in clinical effectiveness, meaning that switching therapies may result in changes in treatment response (10, 11). Even short-term changes in treatment may have meaningful implications for patients as they navigate the impact of formulary decisions.
This study has several limitations. Dispense data may not capture all medication fills until a clinical encounter has occurred. The switching analysis did not evaluate discontinuation, which may underestimate the full extent of treatment changes. Prescribing data reflect clinician intent but do not confirm whether prescriptions were filled. We also did not include insurance class in this analysis; patients in this analysis were likely not affected by the CVS formulary change. Finally, while the timing of observed changes aligns closely with the formulary update, this analysis does not establish causality, and other factors such as regional policies, supply dynamics, or provider preferences may have contributed.
These findings highlight the central role of formulary and coverage decisions in shaping treatment access and underscore the importance of continued monitoring as the GLP-1 therapeutic landscape evolves.
These findings are consistent with data accessed on March 20, 2026.
Citations
- D. J. Drucker, Efficacy and safety of GLP-1 medicines for type 2 diabetes and obesity. Diabetes care 47, 1873–1888 (2024).
- S. Patel, S. K. Niazi, Emerging Frontiers in GLP-1 Therapeutics: A Comprehensive Evidence Base (2025). Pharmaceutics 17, 1036 (2025).
- E. D. Michos, F. Lopez‐Jimenez, M. Gulati, Role of Glucagon‐Like Peptide‐1 Receptor Agonists in Achieving Weight Loss and Improving Cardiovascular Outcomes in People With Overweight and Obesity. JAHA 12, e029282 (2023).
- Monitoring Report: GLP-1 RA Prescribing Trends – December 2025 Data | medRxiv. https://www.medrxiv.org/content/10.1101/2025.03.06.25323524v5.
- Diabetes Patients Also Struggle to Access GLP-1 Agonists | MedPage Today. https://www.medpagetoday.com/special-reports/features/102773.
- J. H. Hwang, N. Laiteerapong, E. S. Huang, D. Mozaffarian, A. M. Fendrick, D. D. Kim, “Fiscal impact of expanded Medicare coverage for GLP-1 receptor agonists to treat obesity” in JAMA Health Forum (2025; https://jamanetwork.com/journals/jama-health-forum/article-abstract/2833038)vol. 6, p. e250905.
- CVS Health Announces It Will Drop Zepbound and Prioritize Coverage of Wegovy. https://www.pharmacytimes.com/view/cvs-health-announces-it-will-drop-zepbound-instead-prioritizing-coverage-of-wegovy.
- F. Kansteiner, In major blow for Lilly, Novo strikes deal giving Wegovy preferred access on CVS Caremark formulary, Fierce Pharma (2025). https://www.fiercepharma.com/pharma/major-blow-lilly-novo-strikes-deal-giving-wegovy-preferred-access-cvs-caremark-formulary.



