Authors: Brianna M Cartwright ⊕Truveta, Inc, Bellevue, WA, Duy Do ⊕Truveta, Inc, Bellevue, WA, Jennifer J Liang ⊕Truveta, Inc, Bellevue, WA, Natasha Geogy ⊕Truveta, Inc, Bellevue, WA, Nina B Masters ⊕Truveta, Inc, Bellevue, WA, Karen G Farrar ⊕Truveta, Inc, Bellevue, WA
- Nearly two-thirds of patients initiating lecanemab were adherent across the first six months of therapy.
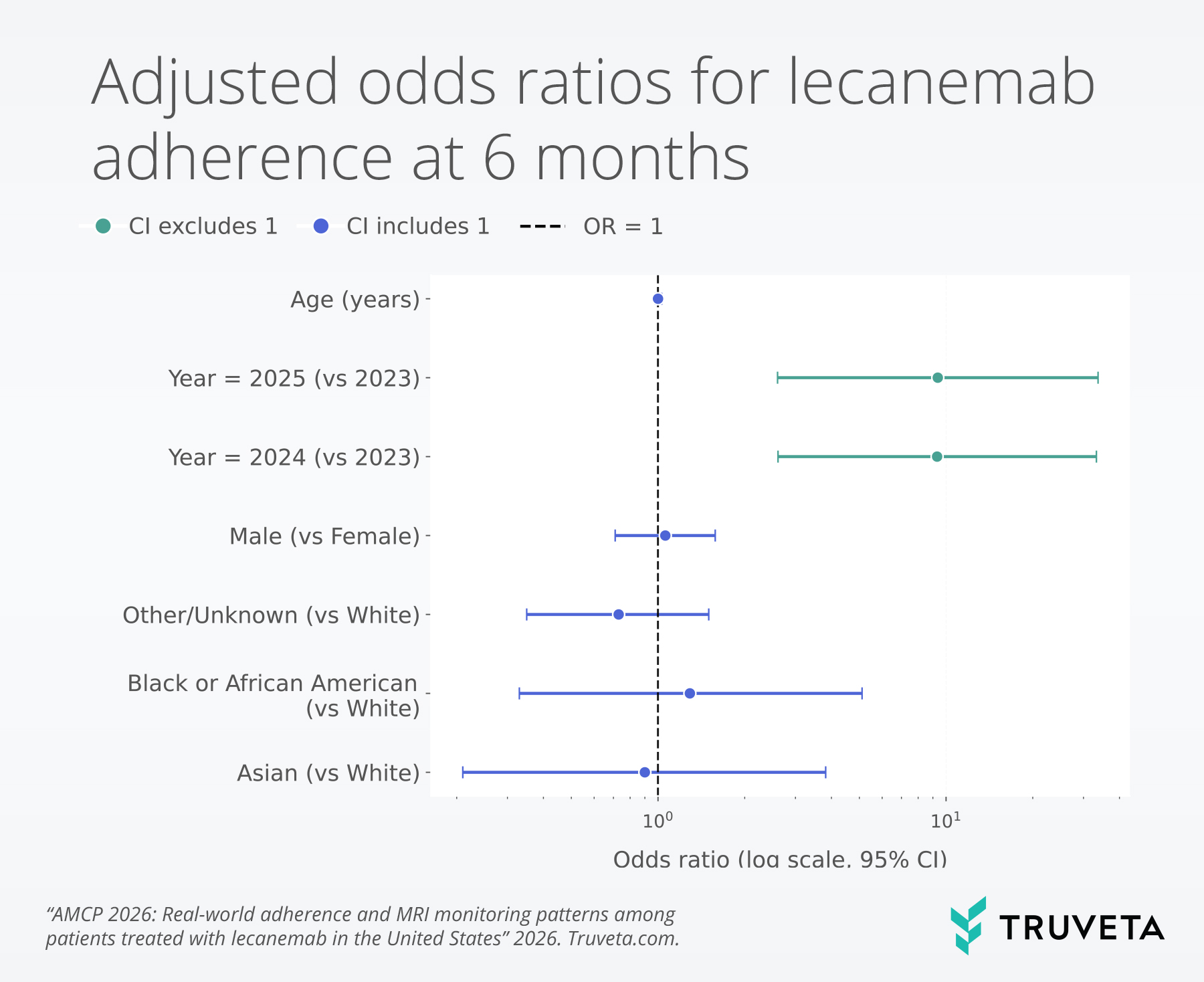
- Adherence increased significantly over time: from 16.7% in 2023 to 64.5% in 2025.
- Most patients (90.6%) received follow-up MRI monitoring consistent with safety recommendations, though small gaps remain.
This blog is an extension of our poster presented at AMCP, titled Real-world adherence and MRI monitoring patterns among patients treated with lecanemab in the United States.
Alzheimer’s disease, the most common cause of dementia, remains a pressing public health challenge (1). The disease affects millions of adults in the United States and contributes substantially to morbidity, mortality, and healthcare . This burden is expected to grow, with the number of people with Alzheimer’s disease projected to double by 2060 (3). Until recently, treatment options were limited to symptomatic therapies, , such as cholinesterase inhibitors and memantine, which do not modify underlying disease progression (4). The approval of disease-modifying therapies targeting amyloid-beta has marked a major shift in the treatment landscape, offering the potential to slow disease progression in patients with early Alzheimer’s disease or mild cognitive impairment (1).
Lecanemab (Leqembi), an anti-amyloid monoclonal antibody, received accelerated FDA approval in January 2023 (5) and full approval in July 2023 for patients with early Alzheimer’s disease (6). Clinical trials demonstrated that lecanemab reduces amyloid burden and modestly slows cognitive decline (7); however, its real-world implementation introduces new challenges. Treatment requires biweekly intravenous infusions and carries a risk of amyloid-related imaging abnormalities (ARIA), necessitating regular MRI monitoring (8). These logistical and safety considerations, along with high treatment costs (9), raise important questions about real-world adherence and feasibility for patients.
Building on prior Truveta research examining early adoption and prescribing patterns of lecanemab, we sought to understand how this therapy is being used in routine clinical practice (10). Specifically, we evaluated six-month adherence, factors associated with adherence, and MRI monitoring patterns among patients initiating lecanemab in real-world settings.
Methods
Using a subset of Truveta Data, we identified patients who initiated lecanemab (with of a medication administration) between January 2023 and September 2025. We expect most patients would not begin treatment until it received the full FDA approval in July 2023. Patients were included in the analysis at the time of treatment initiation. The time-adjusted proportion of patients initiating treatment each month was calculated as the number of patients initiating in a given month divided by the number of patients initiating over the entire study period.
Baseline cognitive assessment
Baseline cognitive function was assessed using the Montreal Cognitive Assessment (MoCA) and Mini-Mental State Examination (MMSE), extracted from clinical notes prior to treatment initiation.
Adherence
Adherence was defined as receiving at least 90% of recommended doses within six months (≥12 of 13 infusions). We examined overall adherence and evaluated predictors of adherence using logistic regression, adjusting for demographic and clinical characteristics.
MRI monitoring
Among patients with available imaging data, we assessed receipt and timing of MRI within six months of treatment initiation to evaluate alignment with safety monitoring recommendations for ARIA.
Results
We identified 458 patients who initiated lecanemab during the study period. The population was older (mean age 73.7 ± 7.0 years), predominantly female (57.4%), and largely White (88.9%) and non-Hispanic (91.7%).
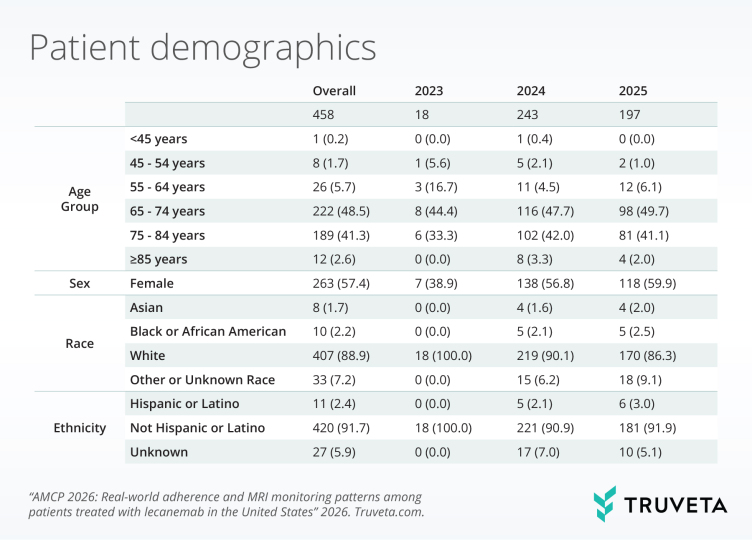
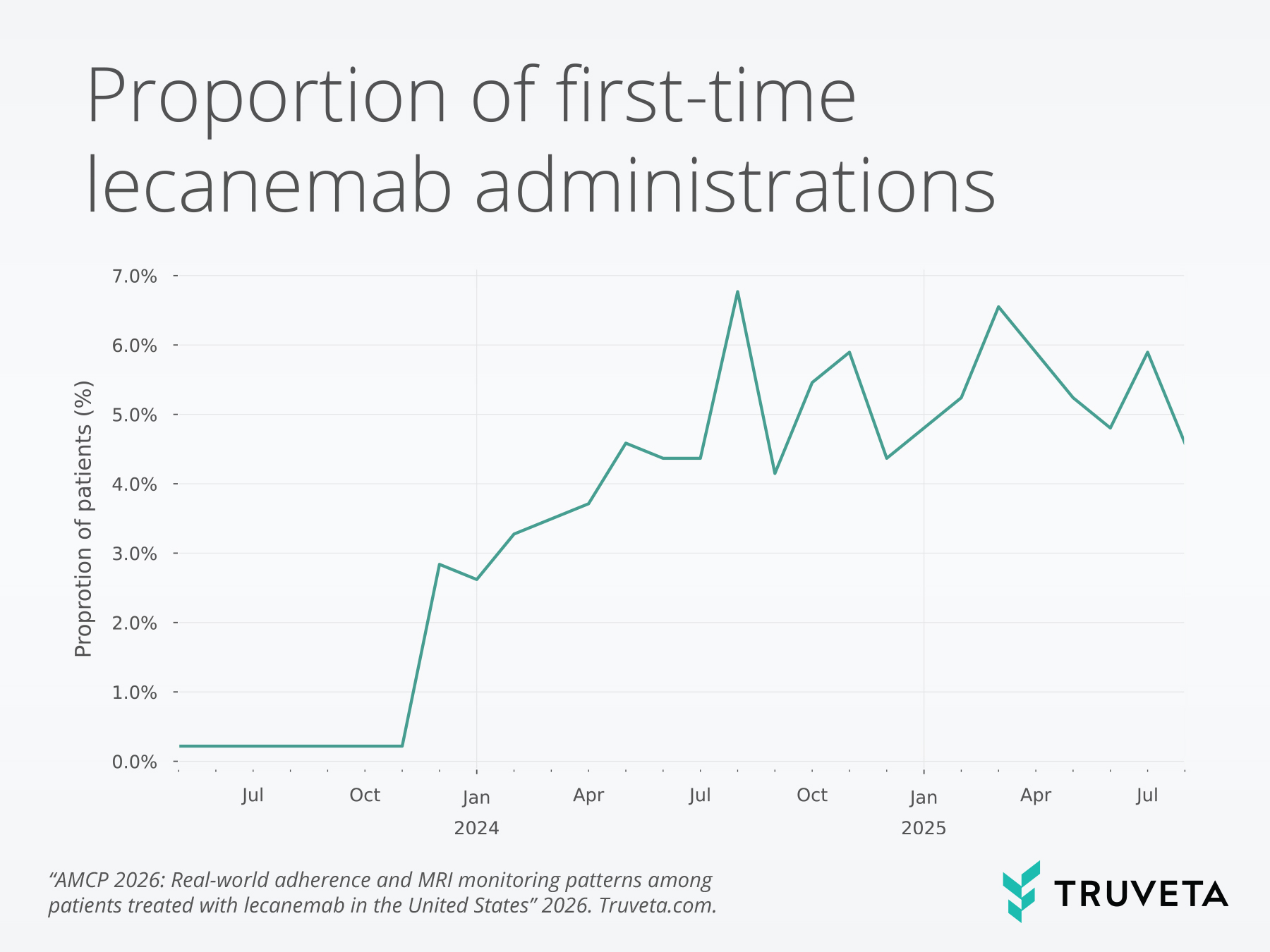
Lecanemab initiation began to increase at the end of 2023, with a greater proportion of patients initiating the medication in 2024 and 2025.
Nearly half of patients (46.5%) had a documented baseline cognitive assessment (either the MoCA or MMSE test). Among those with available data, the mean MoCA score was 20.8 (SD 4.1) and the mean MMSE score was 24.6 (SD 6.2), consistent with mild cognitive impairment or early-stage dementia.
Adherence to lecanemab
Overall, nearly two-thirds (62.7%) of patients were adherent to lecanemab during the first six months of treatment.
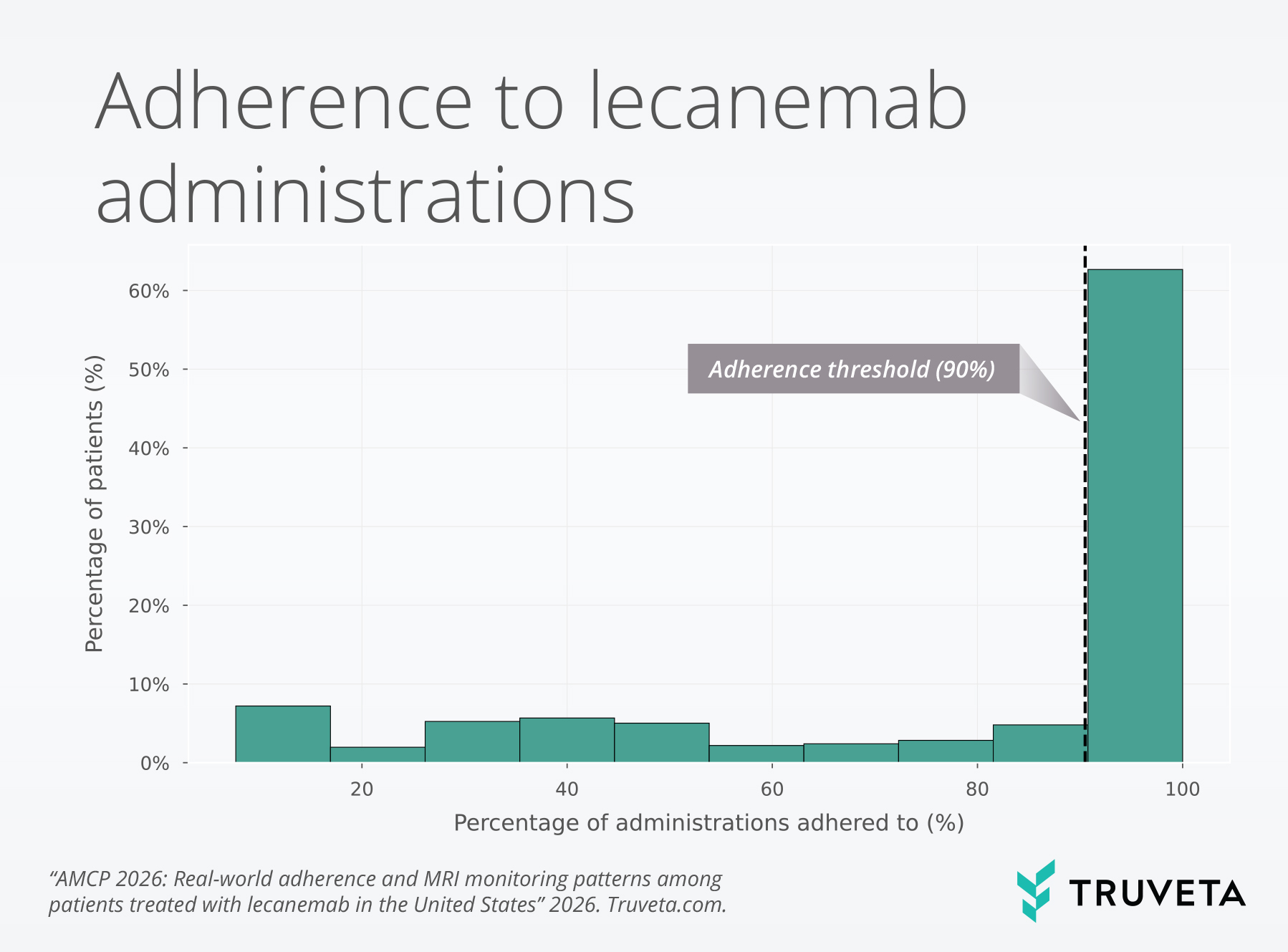
Adherence improved substantially over time, with patients initiating treatment in later years more likely to meet adherence thresholds than those initiating in 2023.
A small proportion of patients discontinued early (7.2%), receiving only one or two doses. Among those who were non-adherent, most discontinued after receiving four or five doses (10.9% of all patients), suggesting drop-off early in the treatment course rather than gradual attrition.
MRI monitoring patterns
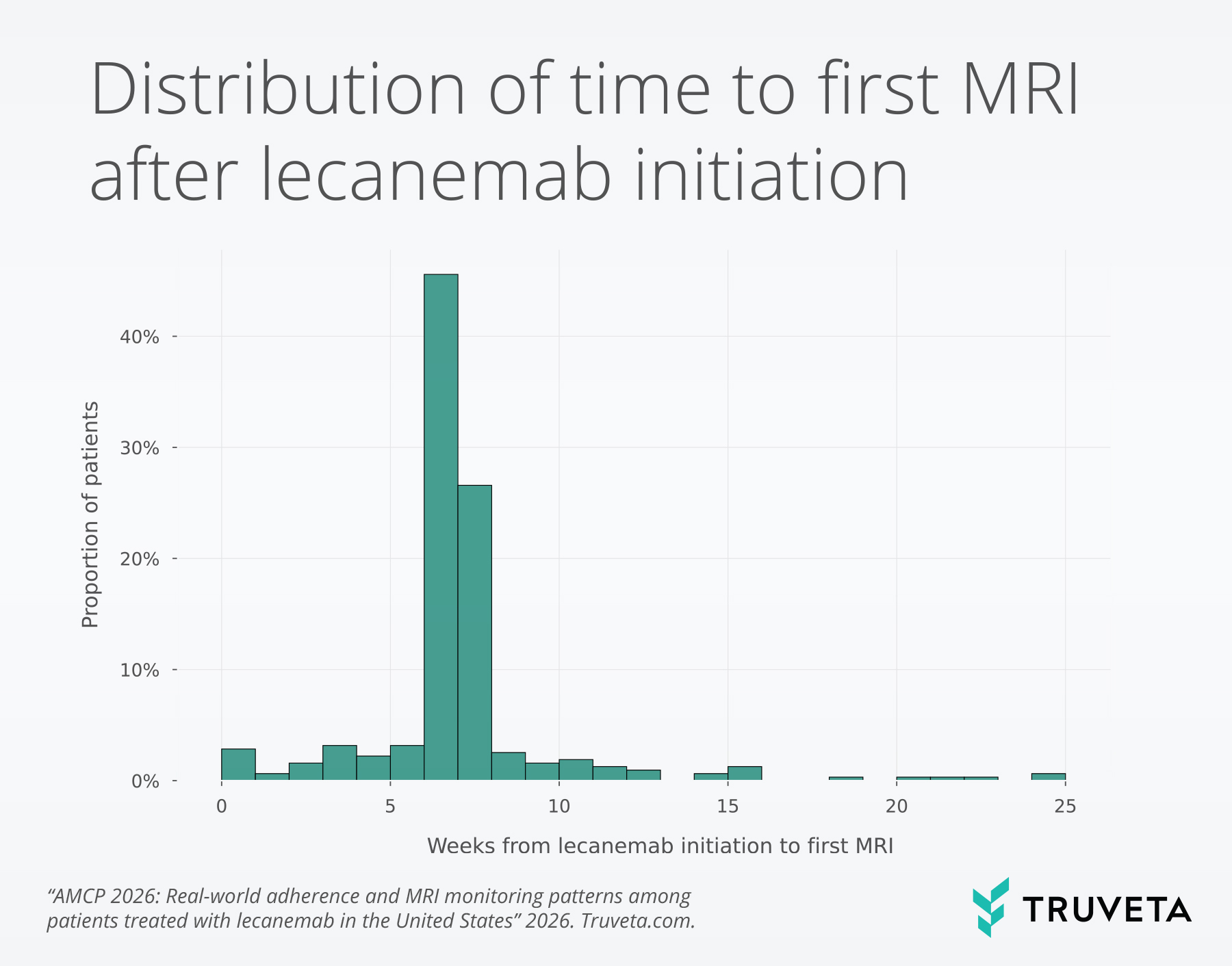
Among patients with prior MRI data (N=341), the vast majority (90.6%) received follow-up imaging within six months of treatment initiation. MRI monitoring was typically conducted after the third dose, aligning with earlier safety monitoring recommendations.
However, approximately 1 in 10 patients did not have documented follow-up MRI within six months, indicating potential gaps in recommended safety monitoring.
Discussion
In this large real-world analysis, we found that nearly two-thirds of patients initiating lecanemab were adherent to treatment over the first six months. Importantly, adherence increased markedly over time, suggesting that providers and health systems are rapidly adapting to the logistical and clinical requirements of this novel therapy. As clinical familiarity grows and care pathways become more standardized, adherence may continue to improve.
These findings are notable given the complexity of lecanemab treatment. Biweekly infusions, monitoring requirements, and potential adverse effects present meaningful barriers to sustained therapy (8). Early concerns about real-world feasibility appear to be partially mitigated by evolving clinical workflows and increased experience with treatment administration. Our findings extend prior Truveta analyses of early lecanemab adoption by demonstrating that not only is uptake occurring and increasing as providers become more familiar with this class of medication, but adherence in practice is higher than might have been expected for a therapy with this level of complexity.
MRI monitoring rates were also high, with most patients receiving follow-up imaging consistent with safety guidance. This suggests strong alignment with recommendations for ARIA surveillance in routine care and aligns with prior literature (11). However, the absence of follow-up MRI in approximately 10% of patients highlights an important area for improvement, particularly given the clinical significance of ARIA detection. Notably, most patients in this study initiated treatment prior to updated FDA guidance recommending earlier MRI monitoring in August 2025 (12), which may further influence future patterns.
Several limitations should be considered. First, EHR data may not capture all infusions or imaging performed outside participating health systems, potentially underestimating adherence or MRI monitoring. Second, we did not require a documented last encounter to ensure adequate follow-up time, which may result in incomplete capture of ongoing care or adherence. Third, cognitive assessments were only available for a subset of patients, as these measures are often recorded in unstructured clinical notes. Fourth, while we identified associations with adherence, causal relationships cannot be established. Finally, as this therapy continues to evolve, longer-term adherence and outcomes remain important areas for future study.
Despite these limitations, this analysis provides important early evidence on the real-world use of lecanemab. The observed improvements in adherence over time and high rates of MRI monitoring suggest that health systems are adapting to support the safe and effective use of disease-modifying therapies for Alzheimer’s disease. Continued evaluation will be critical as treatment guidelines evolve and as more patients initiate therapy.
These findings are consistent with data accessed on March 19, 2026.
Citations
- US Centers for Disease Control and Prevention, About Alzheimer’s (2024). https://www.cdc.gov/alzheimers-dementia/about/alzheimers.html.
- Alzheimer’s Association, “2025 Alzheimer’s Disease Facts and Figures” (2025); https://www.alz.org/getmedia/ef8f48f9-ad36-48ea-87f9-b74034635c1e/alzheimers-facts-and-figures.pdf.
- K. B. Rajan, J. Weuve, L. L. Barnes, E. A. McAninch, R. S. Wilson, D. A. Evans, Population estimate of people with clinical Alzheimer’s disease and mild cognitive impairment in the United States (2020–2060). Alzheimer’s & Dementia 17, 1966–1975 (2021).
- National Institute on Aging, How Is Alzheimer’s Disease Treated? (2023). https://www.nia.nih.gov/health/alzheimers-treatment/how-alzheimers-disease-treated.
- US Food & Drug Administration, FDA Grants Accelerated Approval for Alzheimer’s Disease Treatment. U.S. Food & Drug Administration (2023).
- US Food & Drug Administration, FDA Converts Novel Alzheimer’s Disease Treatment to Traditional Approval (2023). https://www.fda.gov/news-events/press-announcements/fda-converts-novel-alzheimers-disease-treatment-traditional-approval.
- C. H. van Dyck, C. J. Swanson, P. Aisen, R. J. Bateman, C. Chen, M. Gee, M. Kanekiyo, D. Li, L. Reyderman, S. Cohen, L. Froelich, S. Katayama, M. Sabbagh, B. Vellas, D. Watson, S. Dhadda, M. Irizarry, L. D. Kramer, T. Iwatsubo, Lecanemab in Early Alzheimer’s Disease. New England Journal of Medicine 388, 9–21 (2023).
- Eisai & Biogen, Leqembi (lecanemab-imb) (2026). https://www.leqembihcp.com/dosing.
- Alzheimer’s Association, Lecanemab Approved for Treatment of Early Alzheimer’s Disease (2024). https://www.alz.org/alzheimers-dementia/treatments/lecanemab-leqembi#:~:text=How%20much%20will%20this%20drug,for%20financial%20information%20and%20support.
- B. Cartwright, P. Rodriguez, S. Gratzl, C. Baker, N. Stucky, “Real-World Prescribing of Lecanemab in the United States” (Value in Health, 2024; https://www.ispor.org/heor-resources/presentations-database/presentation/intl2024-3895/139682)vol. 27 of 6, S1.
- M. N. Sabbagh, C. Zhao, M. Mahendran, S. R. Jang, F. Laliberté, H. Toyosaki, K. Zhang, F. Frech, K. V. Nair, Characterizing the Journey of Early Alzheimer’s Disease in Patients Initiating Lecanemab Treatment in the United States: A Real-World Evidence Study. Neurol Ther 14, 1115–1127 (2025).
- U.S. Food & Drug Administration, FDA to recommend additional, earlier MRI monitoring for patients with Alzheimer’s disease taking Leqembi (lecanemab) (2025). https://www.fda.gov/drugs/drug-safety-and-availability/fda-recommend-additional-earlier-mri-monitoring-patients-alzheimers-disease-taking-leqembi-lecanemab.



