- Estrogen-based hormone replacement therapy (HRT) prescribing more than doubled from 2018 to 2026.
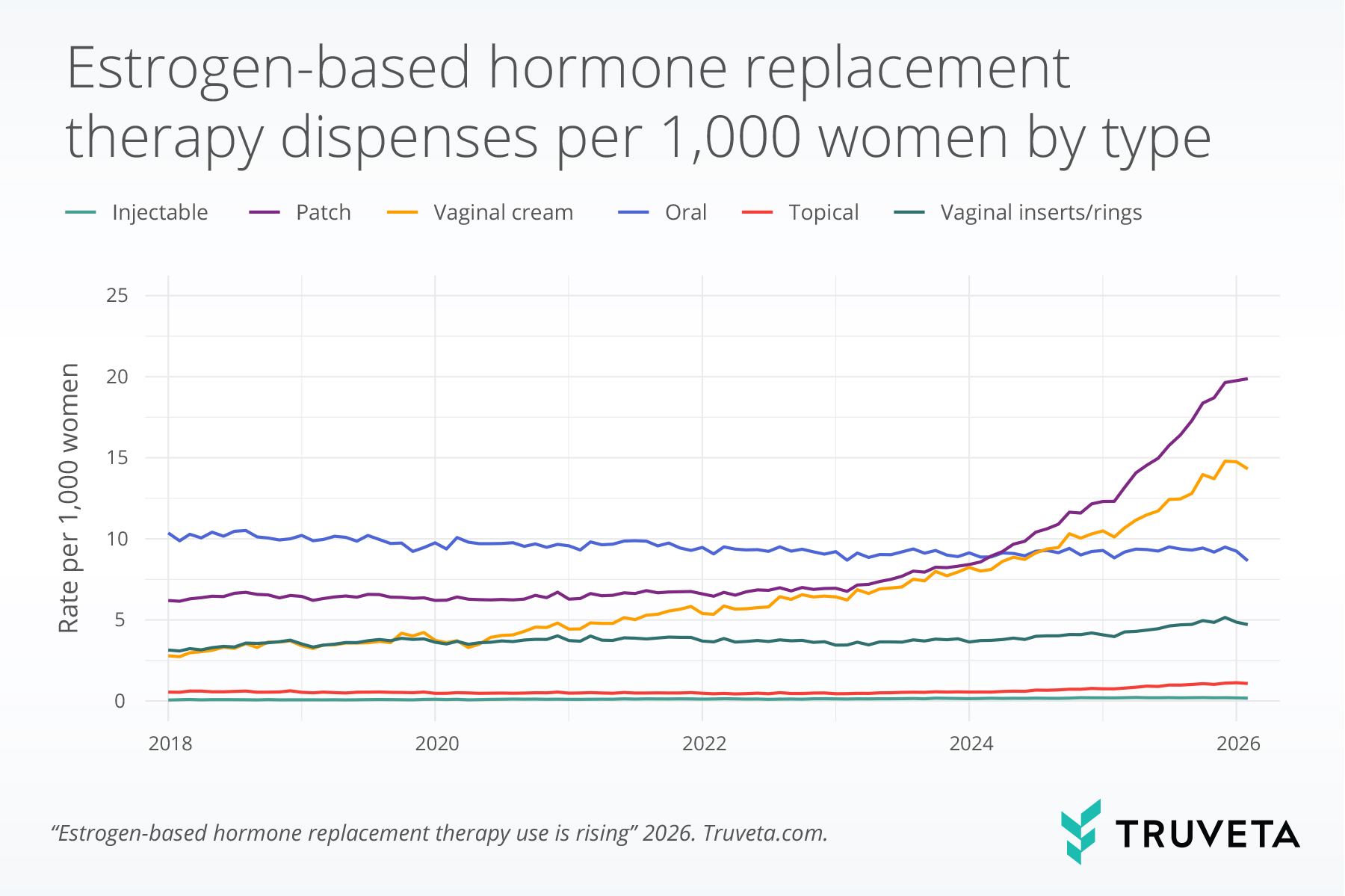
- The largest increases were seen in patches and vaginal creams, with patch use more than tripling and vaginal cream use increasing more than fourfold.
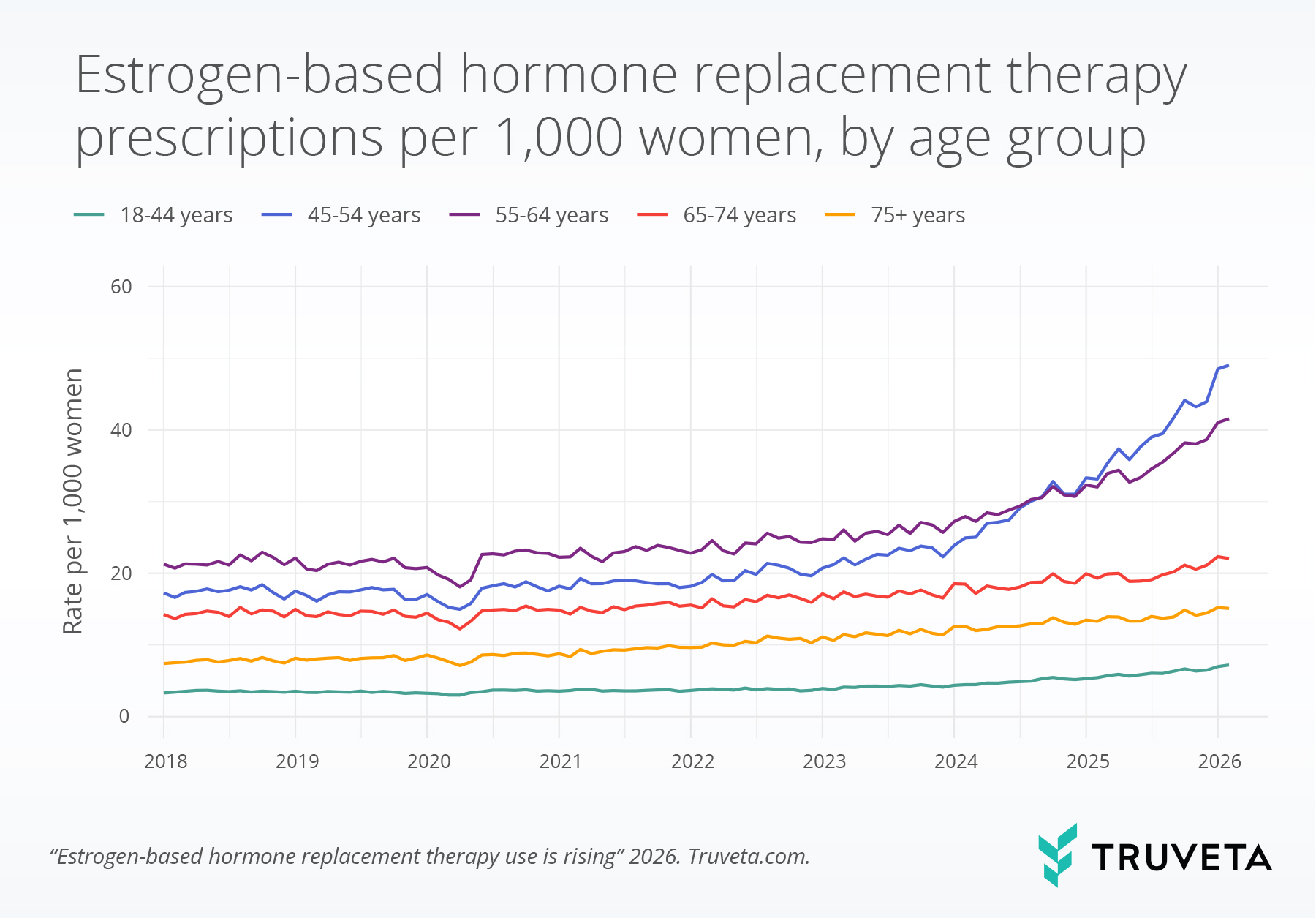
- The rates of women aged 45-54 receiving estrogen-based HRT therapies increased by 184.2% across the study period; in February 2026, 1 in 20 women in this age group had evidence of an estrogen-based HRT prescription.
Menopause and perimenopause affect many aspects of a woman’s health, including physical symptoms, mental health, and cardiometabolic and bone health, with impacts that extend to overall well-being and long-term health (1–3). Hormone replacement therapy (HRT) is a treatment for menopause symptoms like hot flashes, vaginal dryness, and bone loss by supplementing declining levels of estrogen and progesterone that occur during menopause (4, 5). These symptoms can significantly affect quality of life, including sleep, daily functioning, and long-term health, making effective treatment an important component of women’s healthcare (3, 6). However, HRT use decreased sharply in the early 2000s following the Women’s Health Initiative (WHI) 2002 findings raised concerns about cardiovascular and cancer risks associated with certain hormone therapies (1, 2). While the WHI was a landmark study, subsequent analyses have highlighted important limitations in its interpretation, particularly with respect to differences by age, time since menopause, and hormone formulation, which were not fully reflected in the original report (9, 10). Over the past two decades, an accumulating body of evidence has reshaped our understanding of HRT, demonstrating that it is effective for symptom relief and that risks are low for many women, especially those who initiate therapy near the onset of menopause (4, 5, 11, 12). Importantly, both benefits and risks vary by age, timing of initiation, formulation, and duration of use, underscoring the need for a more individualized approach to treatment (1, 4, 9, 10, 13–15).
Building on this evolving evidence base, recent years have also seen rapid changes in the public conversation around menopause care. Public figures including Oprah Winfrey and Michelle Obama, among others, have shared their personal experiences with menopause in an effort to bring greater visibility to a topic that has been historically ignored (16, 17). In parallel, clinicians such as Dr. Mary Claire Haver and Dr. Sharon Malone have further expanded awareness through widely read books and media appearances (18, 19). Together, these efforts have contributed to a broader cultural shift, increasing awareness of menopausal symptoms and available therapies and encouraging more women to seek care.
This renewed attention is also reflected in evolving regulatory guidelines. In July 2025, the US Food and Drug Administration (FDA) convened an expert panel to reassess HRT safety in light of the emerging evidence. The review included the longstanding black box warning, the FDA’s most prominent safety warning, highlighting potential risks such as cardiovascular events and certain cancers (20). Following this review, on November 10, 2025, the FDA removed the black box warnings from certain hormone therapy products, reflecting updated evidence on risks and benefits (21). At the same time, recent reports of shortages in estrogen patches have raised additional questions about access and prescribing patterns for these therapies (22, 23). Together, these regulatory and societal shifts are likely influencing both clinicians prescribing behavior and patient demand for HRT.
In this study, we examined recent trends in estrogen-based HRT use using a subset of Truveta Data. Given recent questions around availability of estrogen patches, we focused specifically on estrogen-based HRT (22, 23). We evaluated prescribing rates over time and dispensing patterns by route of administration since 2018, with particular attention to changes following the July 2025 FDA expert panel, to understand how utilization has evolved across age groups and treatment types.
Methods
Using a subset of Truveta Data, we evaluated trends in estrogen-based HRT using two complementary cohorts.
Prescribing trends
We examined monthly prescribing rates of estrogen-based HRT among women aged 18 years and older with available prescription data from 2018 to February 2026. Estrogen-based HRT prescribing rates were defined as the number of women with an estrogen-based HRT prescription per 1,000 women with prescription data. We also examined rates by age group.
Dispensing trends by route of administration
We examined monthly dispensing rates of estrogen-based HRT by route of administration among women aged 18 years and older with available dispensing data from 2018 to February 2026. Patients in this cohort were also required to have at least one healthcare encounter in 2024, 2025, or 2026. Dispensing rates were defined as the number of women with an estrogen-based HRT medication dispense (prescription filled at a pharmacy) per 1,000 women with dispensing data, stratified by formulation type (oral, patch, vaginal cream, vaginal insert/ring, topical formulations [including gels, sprays, and creams], and injectable).
Results
Prescribing trends
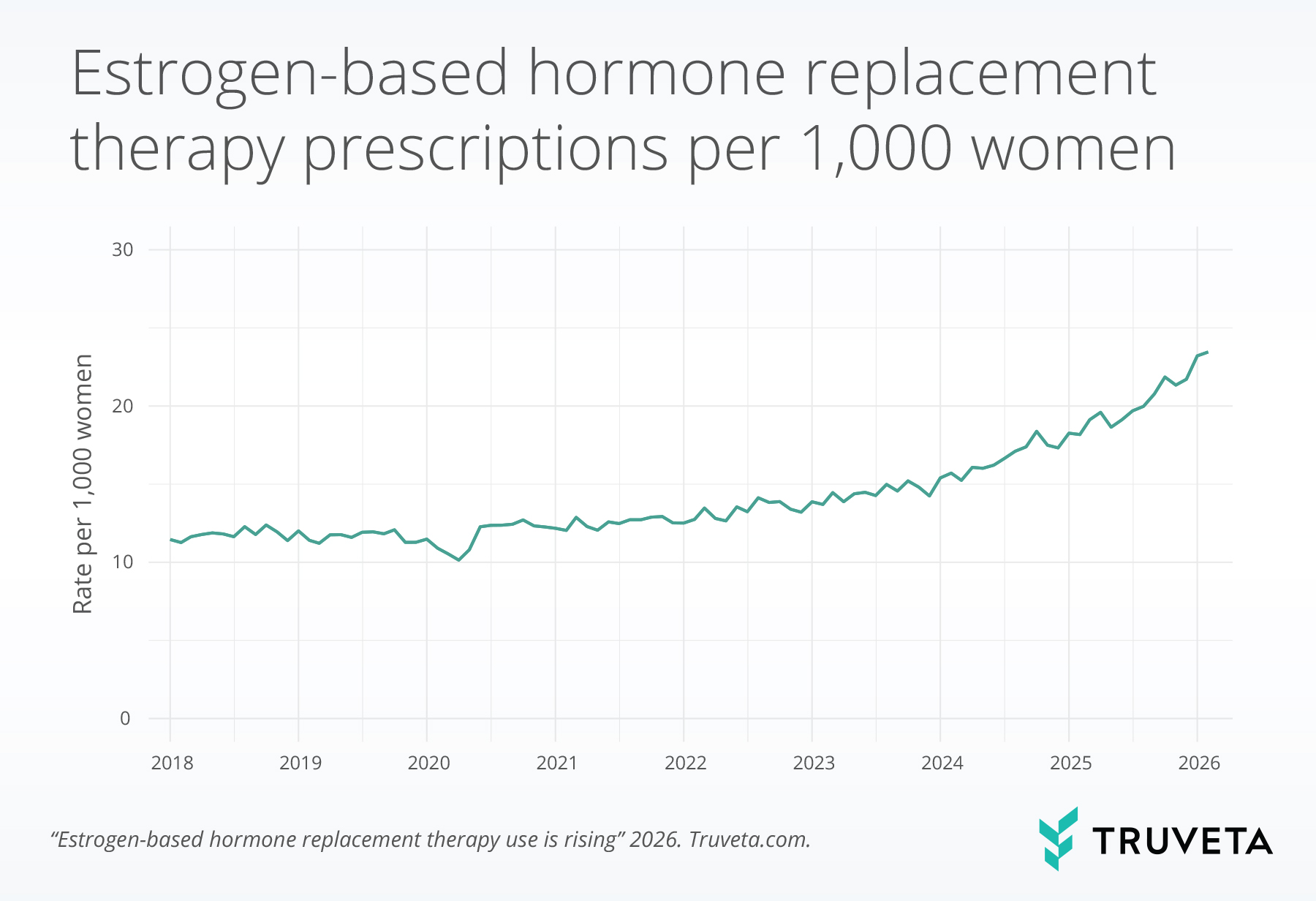
Estrogen-based HRT prescribing increased substantially over the study period, rising from 11.5 per 1,000 women in January 2018 to 23.5 per 1,000 women in February 2026, a 104.8% increase. This upward trend persisted in recent months, with rates increasing 19.1% between July 2025 (19.7 per 1,000) and February 2026. However, overall use remained low, with only about 2 in 100 women having evidence of estrogen-based HRT use.
Prescribing trends by age
Estrogen-based HRT prescribing increased across all age groups over the study period, with the highest rates observed among women aged 45-54 at the end of the study period. By February 2026, rates reached 49.0 per 1,000 in this age group (184.2% increase from 2018), followed by 41.6 per 1,000 among those aged 55–64 (95.3% increase) who had the highest rates at the beginning of the study period. Lower rates were seen for the youngest and oldest age groups; 7.2 per 1,000 women among those aged 18–44 (118.3% increase from 2018), 22.1 per 1,000 among those aged 65–74 (54.8% increase), and 15.1 per 1,000 among those aged 75 and older (103.9% increase).
This upward trend persisted in recent months, with rates increasing from July 2025 most predominantly for those from 45-64 years old. Between July 2025 and February 2026, the rate of prescriptions increased by 25.7% among those aged 45–54, compared with 20.3% among those aged 55–64. All younger and older age groups at lower rates of growth across this time period.
Dispensing trends by route of administration
Estrogen-based HRT dispensing trends varied by route of administration over the study period. While estrogen-based patches were the second most common route of administration in 2018, rates more than tripled across the study period (increasing from 6.2 to 19.9 per 1,000 women), making the patch the most common form of administration in February 2026. Across the study period vaginal creams showed the largest relative increase, rising from 2.8 to 14.3 per 1,000 (413.3% increase). Although vaginal inserts/rings and topical formulations increased it was at a more modest rate, from 3.1 to 4.7 per 1,000 (50.0% increase) and 0.5 to 1.0 per 1,000 (97.2% increase), respectively. Injectable use remained low, but doubled across the study period, increasing from 0.1 to 0.2 per 1,000 (186.3% increase). In contrast, oral HRT use declined from 10.4 to 8.6 per 1,000 (16.5% decrease).
We have continued to see rates increase in recent months. From July 2025 to February 2026, patch use increased by 26.1% and vaginal cream by 15.2%, while topical formulations rose by 9.5%. Vaginal inserts or rings remained relatively stable (+1.9%). In contrast, oral HRT use declined further (−9.0%), and injectable use also decreased (−16.2%).
Discussion
In this analysis of estrogen-based HRT use from 2018 to early 2026, we observed a substantial and sustained increase in prescribing rates, alongside notable shifts in the types of therapies being dispensed. Estrogen-based HRT prescribing more than doubled over the study period and continued to rise in recent months after the FDA expert panel on HRT safety in July 2025.
These findings align with a broader resurgence of interest in menopause care. Increased public awareness, driven by media coverage, educational campaigns, and advocacy from clinicians and public figures, may be contributing to greater recognition of menopausal symptoms and available treatment options (16–19). The FDA’s 2025 decision to remove longstanding boxed warnings from certain hormone therapies further reflects this evolving understanding and may reduce barriers to prescribing (20, 21).
In our analysis, rates of oral estrogen-based HRT dispensing declined over the study period, while rates of patches and vaginal creams dispensing increased substantially, with patch use more than tripling and vaginal cream use increasing more than fourfold. The shift in dispensing patterns from oral to transdermal and vaginal formulations is consistent with evolving clinical guidance (24, 25). Some observational evidence suggests that transdermal therapies may be associated with lower risk of certain side effects compared to oral formulations (1, 24–26). Together, these trends suggest that clinicians are not only prescribing HRT more frequently, but also selecting different routes of administration.
We also observed meaningful differences in HRT prescribing across age groups. While increases were seen in all age categories, the largest relative growth occurred among women aged 45–54 years, the population most likely to be experiencing menopausal transition. By early 2026, prescribing rates in this group reached nearly 5%, meaning 1 in 20 women in this age group received an estrogen-based HRT prescription. This pattern suggests that rising estrogen-based HRT use is being driven in part by greater recognition and treatment of perimenopausal and early menopausal symptoms (1).
Despite reports of shortages affecting certain estrogen patch products in recent months (22, 23, 27), we did not observe a corresponding decline in patch dispensing in our data. Additionally, there has been no shortage announcement from the FDA during this period (28). It is possible that any emerging supply disruptions may not yet be reflected in prescribing or dispensing patterns. For example, estrogen patches are often dispensed with 30- or 90-day supplies, which could delay observable impacts in dispensing data. Patients may also switch between brands, travel farther or use different pharmacies to obtain medications. Continued monitoring will be important to assess whether supply dynamics influence future utilization trends.
This study has several limitations. First, our analysis focused on estrogen-based HRT and does not capture all treatment for menopausal symptoms, including progesterone-only therapies, non-hormonal therapies such as selective serotonin reuptake inhibitors (SSRIs) or hormonal contraceptives (e.g., oral contraceptives or intrauterine devices), which may also be used to treat menopausal symptoms. Second, we did not confirm menopausal status, and some prescriptions may reflect use for other indications. Third, while we observed increases in prescribing, we did not directly assess whether prescriptions were filled or adherence to therapy. Finally, while we did not observe a noticeable decline in estrogen patch dispensing relative to other formulations, extended days’ supply (e.g., 30- or 90-day fills) could delay the ability to detect changes in utilization or supply.
Our findings highlight a meaningful shift in estrogen-based HRT prescribing and dispensing in recent years. We observed a marked increase in estrogen-based HRT use, with the most pronounced growth seen in patches and vaginal creams. These trends likely reflect greater public recognition of menopausal care and evolving regulatory standards. As use continues to rise, understanding how these therapies are used in real-world settings will be important for understanding how they influence women’s health outcomes.
These are preliminary research findings and not peer reviewed. Data are regularly updating. These findings are consistent with data accessed on March 30, 2026.
Citations
- L. Cho, A. M. Kaunitz, S. S. Faubion, S. N. Hayes, E. S. Lau, N. Pristera, N. Scott, J. L. Shifren, C. L. Shufelt, C. A. Stuenkel, K. J. Lindley, for the ACC CVD in Women Committee, Rethinking Menopausal Hormone Therapy: For Whom, What, When, and How Long? Circulation 147, 597–610 (2023).
- R. Hess, R. C. Thurston, R. D. Hays, C.-C. H. Chang, S. N. Dillon, R. B. Ness, C. L. Bryce, W. N. Kapoor, K. A. Matthews, The impact of menopause on health-related quality of life: results from the STRIDE longitudinal study. Qual Life Res 21, 535–544 (2012).
- J. Whiteley, M. daCosta DiBonaventura, J.-S. Wagner, J. Alvir, S. Shah, The Impact of Menopausal Symptoms on Quality of Life, Productivity, and Economic Outcomes. J Womens Health (Larchmt) 22, 983–990 (2013).
- Y. Tang, R. Ma, L. Zhang, X. Sun, Y. Wang, Effectiveness and safety of hormone replacement therapy in the treatment of menopausal syndrome: a meta-analysis. Am J Transl Res 17, 1–15 (2025).
- C. R. Cameron, S. Cohen, K. Sewell, M. Lee, The Art of Hormone Replacement Therapy (HRT) in Menopause Management. Journal of Pharmacy Practice 37, 736–740 (2024).
- K. L. Hildreth, C. Ozemek, W. M. Kohrt, P. J. Blatchford, K. L. Moreau, Vascular dysfunction across the stages of the menopause transition is associated with menopausal symptoms and quality of life. Menopause 25, 1011–1019 (2018).
- S. L. Crawford, C. J. Crandall, C. A. Derby, S. R. El Khoudary, L. E. Waetjen, M. Fischer, H. Joffe, Menopausal Hormone Therapy Trends Before Versus After 2002: Impact of the Women’s Health Initiative Study Results. Menopause 26, 588–597 (2018).
- Writing Group for the Women’s Health Initiative Investigators, Risks and Benefits of Estrogen Plus Progestin in Healthy Postmenopausal WomenPrincipal Results From the Women’s Health Initiative Randomized Controlled Trial. JAMA 288, 321–333 (2002).
- S. M. Harman, Estrogen replacement in menopausal women: Recent and current prospective studies, the WHI and the KEEPS. Gender Medicine 3, 254–269 (2006).
- R. C. Chester, J. M. Kling, J. E. Manson, What the Women’s Health Initiative has taught us about menopausal hormone therapy. Clin Cardiol 41, 247–252 (2018).
- J. H. Pickar, D. F. Archer, R. Kagan, J. V. Pinkerton, H. S. Taylor, Safety and benefit considerations for menopausal hormone therapy. Expert Opinion on Drug Safety 16, 941–954 (2017).
- S. R. Salpeter, J. M. E. Walsh, E. Greyber, T. M. Ormiston, E. E. Salpeter, Mortality associated with hormone replacement therapy in younger and older women: a meta-analysis. J Gen Intern Med 19, 791–804 (2004).
- M. Hickey, J. Elliott, S. L. Davison, Hormone replacement therapy. Bmj 344 (2012).
- S. Palacios, Advances in hormone replacement therapy: making the menopause manageable. BMC Women’s Health 8, 22 (2008).
- R. D. Langer, H. N. Hodis, R. A. Lobo, M. A. Allison, Hormone replacement therapy – where are we now? Climacteric 24, 3–10 (2021).
- 21 Celebs Get Personal With Their Perimenopause and Menopause Stories. https://www.theflowspace.com/lists/celebrities-perimenopause-menopause-stories/drew-barrymore-50/.
- S. Orgad, K. Gilchrist, C. Rottenberg, How to tame your hormones: menopause rage in media discourse. Feminist Media Studies, 1–20 (2024).
- D. Friedman, The Menopause Queen’s Gambit, The New York Times (2024). https://www.nytimes.com/2024/10/30/well/dr-mary-claire-haver-menopause.html.
- Sharon Malone, MD, Is Putting Women’s Health First in a New Podcast. https://www.oprahdaily.com/life/health/a68001953/sharon-malone-podcast/.
- FDA Expert Panel on Menopause and Hormone Replacement Therapy for Women – 07/17/2025, FDA (2025). https://www.fda.gov/patients/fda-expert-panels/fda-expert-panel-menopause-and-hormone-replacement-therapy-women-07172025.
- FDA, FACT SHEET: FDA Initiates Removal of “Black Box” Warnings from Menopausal Hormone Replacement Therapy Products (2025). https://www.hhs.gov/press-room/fact-sheet-fda-initiates-removal-of-black-box-warnings-from-menopausal-hormone-replacement-therapy-products.html.
- Drug Shortage Detail: Estradiol Transdermal System. https://www.ashp.org/drug-shortages/current-shortages/drug-shortage-detail.aspx?id=1206&loginreturnUrl=SSOCheckOnly.
- Nina Agarwal, Why Estrogen Patches For Menopause Are So Hard to Find – The New York Times. https://www.nytimes.com/2026/02/26/well/estrogen-patches-menopause-treatment.html.
- M. Š. Goldštajn, M. Mikuš, F. A. Ferrari, M. Bosco, S. Uccella, M. Noventa, P. Török, S. Terzic, A. S. Laganà, S. Garzon, Effects of transdermal versus oral hormone replacement therapy in postmenopause: a systematic review. Arch Gynecol Obstet 307, 1727–1745 (2022).
- M. P. Goodman, Are All Estrogens Created Equal? A Review of Oral vs. Transdermal Therapy. Journal of Women’s Health 21, 161–169 (2012).
- Postmenopausal Estrogen Therapy Route of Administration and Risk of Venous Thromboembolism | ACOG. https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2013/04/postmenopausal-estrogen-therapy-route-of-administration-and-risk-of-venous-thromboembolism.
- Allison Aubrey, Why the “mad scramble” to fill hormone therapy prescriptions for menopause, NPR (2026). https://www.npr.org/2026/03/10/nx-s1-5742817/menopause-hormone-therapy-shortages.
- FDA Drug Shortages. https://www.accessdata.fda.gov/scripts/drugshortages/default.cfm.


