- A large proportion of early users (36.1%) of oral semaglutide (Wegovy pill) had no prior evidence of GLP-1-based medication use, suggesting rapid uptake among patients new to taking GLP-1 medications.
- Among patients who previously used a GLP-1-based medication, most switched from injectable anti-obesity medications (AOM), including injectable AOM semaglutide (sold as Wegovy) and AOM tirzepatide (sold as Zepbound).
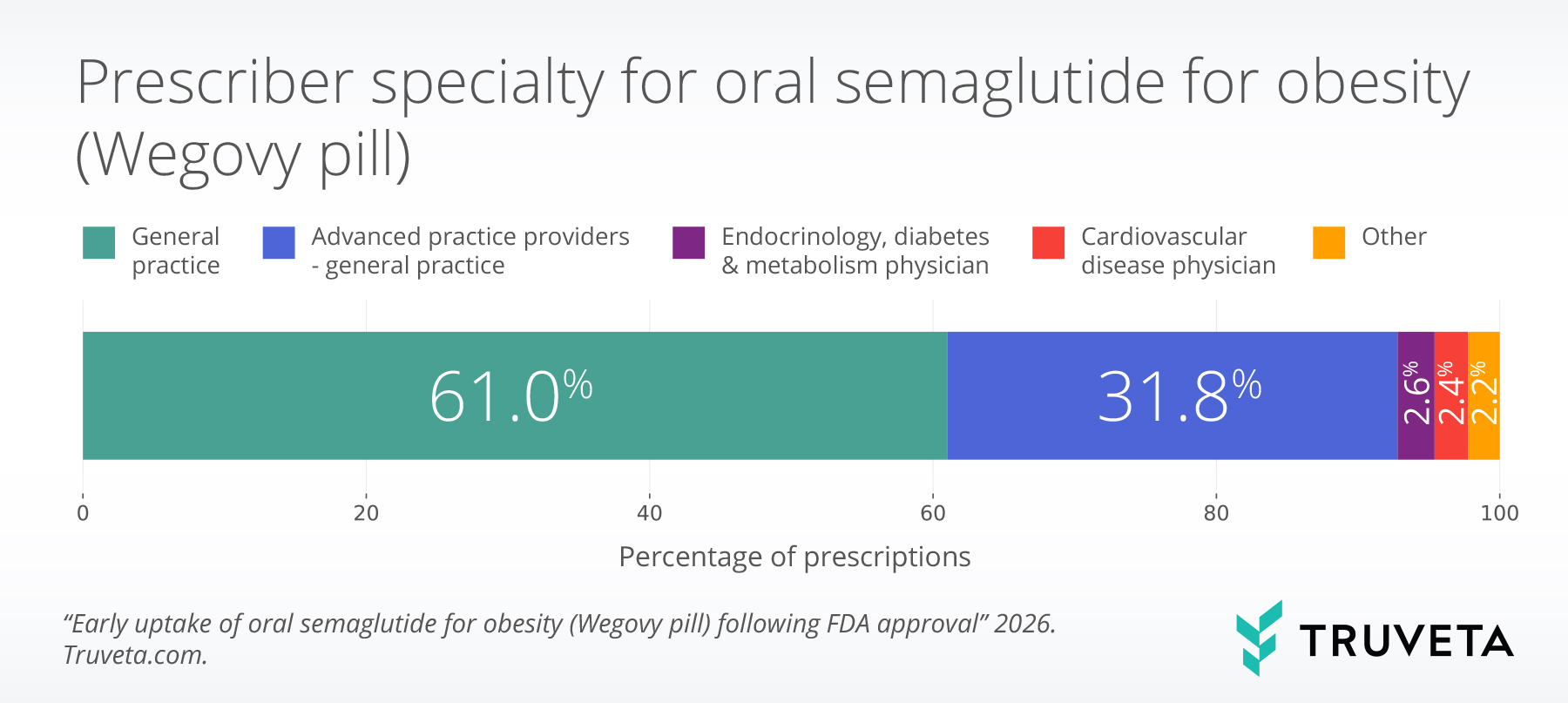
- Most prescriptions were written by general practice providers, highlighting early adoption outside of specialty care.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and GLP-1/GIP dual agonists, collectively referred to as GLP-1-based medications, have transformed the treatment landscape for obesity and cardiometabolic disease. Injectable GLP-1-based therapies, including semaglutide and tirzepatide, have demonstrated substantial benefits for weight loss and cardiometabolic risk reduction (1–3), but real-world uptake has been shaped by factors such as route of administration, access, tolerability, and insurance coverage (4–7). While our recent report has shown that over 7% of all prescriptions are for GLP-1-based therapies (8), oral formulations may lower barriers to initiation by offering a non-injectable option that can be prescribed and managed in a broader range of clinical settings.
On December 22, 2025, the US Food and Drug Administration approved an oral formulation of semaglutide (Wegovy pill) for chronic weight management (9). This approval marked the first oral GLP-1 RA indicated specifically for obesity, creating a new option for patients and clinicians who prefer or require non-injectable therapies, in contrast to Rybelsus, an oral semaglutide approved for the treatment of type 2 diabetes. While injectable GLP-1-based therapy uptake has been well documented (8), far less is known about how quickly newly approved obesity therapies are adopted in routine clinical practice, particularly in the weeks immediately following approval.
Using Truveta Data, we sought to answer a few simple but timely questions: who is receiving the oral semaglutide pill in the first weeks following FDA approval, what provider types are prescribing these drugs, and are these patients new GLP-1-therapy initiators or patients switching from an existing prescription?
Methods
Using a subset of Truveta Data, we identified patients with evidence of either a prescription or a prescription filled at a pharmacy for oral semaglutide between December 22, 2025 (FDA approval date) and February 8, 2026.
We described patient demographics and most recent prior GLP-1-based medication use. Patients were required to have an outpatient encounter in the year prior to the oral semaglutide evidence to be included in the prior GLP-1-based medication pattern analysis.
For patients with available prescribing data, we also examined the specialty of the prescribing clinician. Analyses were descriptive and designed to provide an early look of real-world uptake shortly after market entry.
You can view this study directly in Truveta Studio, including all definitions and codes used in the analysis.
Results
We identified 8,762 patients with evidence of a prescription or prescription fill for oral semaglutide during the first six weeks following FDA approval. Patients were predominantly female (73.3%), White (72.8%), non-Hispanic or Latino (85.8%), and living in urban areas (71.6%). The cohort skewed older, with 34.0% aged 45–59 years and 37.8% aged 60 years or older. Prior evidence of obesity was common (83.9%), and an additional 8.2% had both obesity and type 2 diabetes.
Prior GLP-1-based medication use
The majority of patients (94.2%) had an encounter within the prior year and were therefore included in the switching pattern analysis. Of these patients, a large proportion initiating oral semaglutide (36.1%) had no prior evidence of GLP-1-therapy use, suggesting that early adoption included a substantial proportion of patients new to using GLP-1 medications.
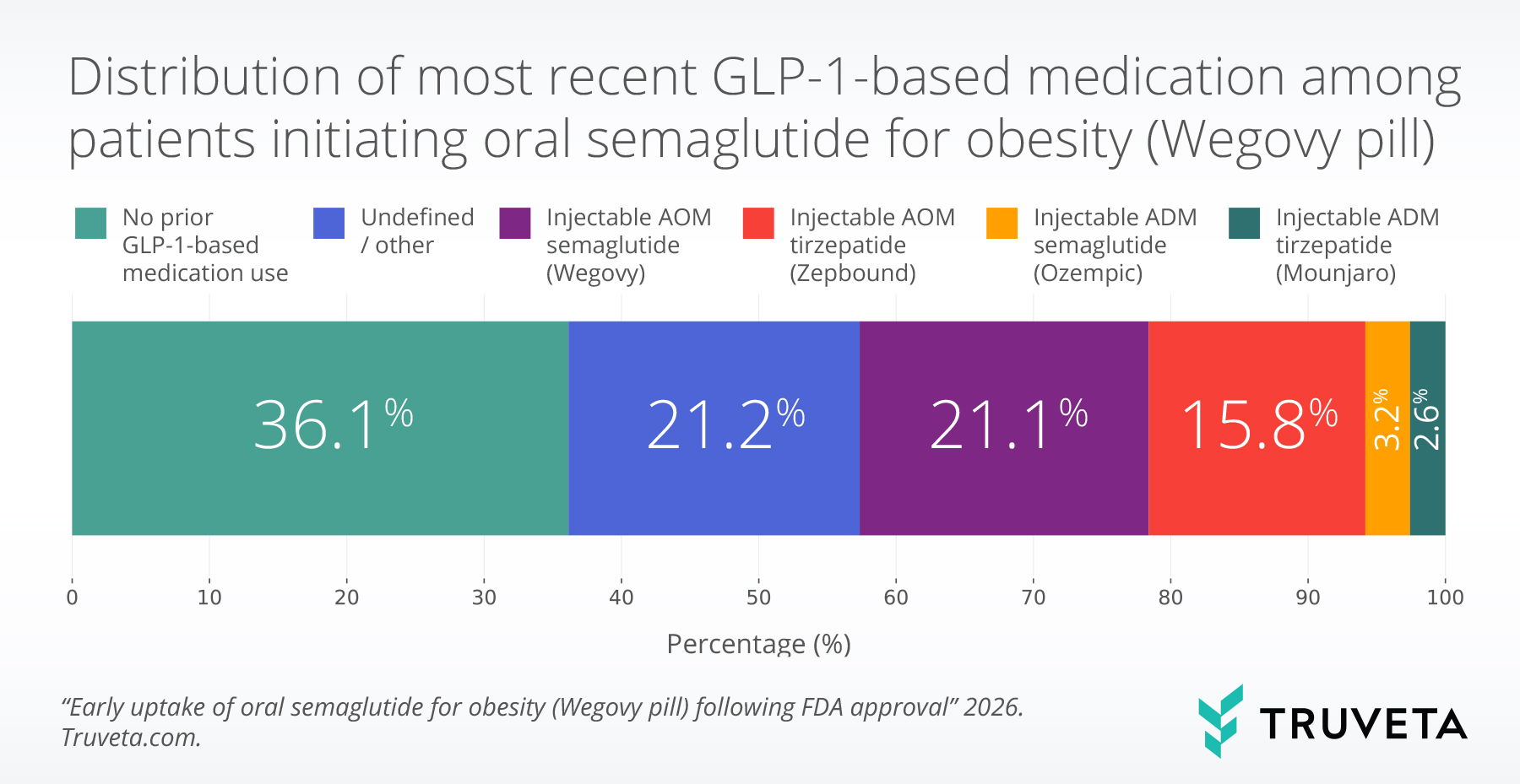
Among patients with evidence of prior GLP-1 therapy, switching from injectable anti-obesity medications was common. Injectable AOM semaglutide accounted for 33.2% of oral semaglutide for obesity initiators, while 24.5% had prior evidence of AOM tirzepatide.
Prescribing clinician specialty
Among patients with available prescribing data (65.2% of the total population), most oral semaglutide prescriptions were written by general practice providers (92.8%). Smaller proportions were prescribed by endocrinology, diabetes, and metabolism specialists (2.6%) and cardiovascular disease specialists (2.3%). This distribution suggests early adoption largely occurred in primary care settings rather than being driven primarily by specialty practices.
Discussion
Within the last six weeks the FDA approved oral semaglutide for obesity and Truveta Data allowed for early characterization of initial prescribing and dispensing patterns. A large proportion had no prior evidence of GLP-1-based medications, highlighting the potential for oral formulations to expand access beyond patients already receiving injectable therapies. At the same time, a substantial proportion of uptake reflected switching from injectable AOM GLP-1-based medications, particularly AOM tirzepatide and injectable AOM semaglutide. This suggests patient or clinician preference for oral administration.
Prescribing patterns further underscore the role of general practice providers in early adoption. The predominance of primary care prescribing suggests that the oral formulation may lower barriers to use outside of specialty care, aligning with its potential to broaden access to obesity treatment. Smaller contributions from cardiovascular and endocrinology specialists may reflect patients with more complex cardiometabolic profiles.
This analysis highlights the ability to rapidly assess early real-world uptake of newly approved therapies within weeks of FDA authorization, enabling timely characterization of initial prescribing patterns as they emerge in clinical practice.
Several limitations should be considered. These findings represent an early view of patients receiving prescriptions or prescription fills of oral semaglutide for obesity. This may not reflect longer-term adoption patterns as payer coverage, prescribing familiarity, and patient demand evolve. For example, we may see increased switching partners as patients need new prescriptions for their existing medications. As additional data accrue, future analyses should examine persistence, switching over time, and clinical outcomes associated with oral semaglutide use.
Despite these limitations, this early analysis provides timely insight into who is receiving oral semaglutide for obesity immediately following approval and demonstrates the value of real-world data for monitoring new drug uptake in near real time.
These are preliminary research findings and not peer reviewed. Data are regularly updating. These findings are consistent with data accessed on February 9, 2026.
Citations
- J. P. H. Wilding, R. L. Batterham, S. Calanna, M. Davies, L. F. Van Gaal, I. Lingvay, B. M. McGowan, J. Rosenstock, M. T. D. Tran, T. A. Wadden, S. Wharton, K. Yokote, N. Zeuthen, R. F. Kushner, Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med 384, 989–1002 (2021).
- A. M. Jastreboff, L. J. Aronne, N. N. Ahmad, S. Wharton, L. Connery, B. Alves, A. Kiyosue, S. Zhang, B. Liu, M. C. Bunck, A. Stefanski, Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med 387, 205–216 (2022).
- S. P. Marso, S. C. Bain, A. Consoli, F. G. Eliaschewitz, E. Jódar, L. A. Leiter, I. Lingvay, J. Rosenstock, J. Seufert, M. L. Warren, V. Woo, O. Hansen, A. G. Holst, J. Pettersson, T. Vilsbøll, Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med 375, 1834–1844 (2016).
- P. J. Rodriguez, V. Zhang, S. Gratzl, D. Do, B. Goodwin Cartwright, C. Baker, T. J. Gluckman, N. Stucky, E. J. Emanuel, Discontinuation and Reinitiation of Dual-Labeled GLP-1 Receptor Agonists Among US Adults With Overweight or Obesity. JAMA Network Open 8, e2457349–e2457349 (2025).
- B. M. G. Cartwright, P. J. Rodriguez, D. Duy, N. Stucky, “Real-World Temporal and Indication-Specific Variation in Drivers of GLP-1 RA Discontinuation” (Value in Health, 2025; https://www.ispor.org/heor-resources/presentations-database/presentation-cti/ispor-2025/poster-session-3/real-world-temporal-and-indication-specific-variation-in-drivers-of-glp-1-ra-discontinuation)vol. 28 of S1.
- A. Montero, G. Sparks, M. Presiado, L. Hamel, KFF Health Tracking Poll May 2024: The Public’s Use and Views of GLP-1 Drugs. Kaiser Family Foundation (2024).
- L. A. Eberly, L. Yang, U. R. Essien, N. D. Eneanya, H. M. Julien, J. Luo, A. S. Nathan, S. A. M. Khatana, E. J. Dayoub, A. C. Fanaroff, J. Giri, P. W. Groeneveld, S. Adusumalli, Racial, Ethnic, and Socioeconomic Inequities in Glucagon-Like Peptide-1 Receptor Agonist Use Among Patients With Diabetes in the US. JAMA Health Forum 2, e214182 (2021).
- S. Gratzl, K. G. Farrar, D. Do, N. B. Masters, B. M. G. Cartwright, Monitoring Report: GLP-1 RA Prescribing Trends – December 2025 Data. [Preprint] (2025). https://doi.org/10.1101/2025.03.06.25323524.
- Novo Nordisk Inc., FDA approves Novo Nordisk’s Wegovy® pill, the first and only oral GLP-1 for weight loss in adults (2025). https://www.prnewswire.com/news-releases/fda-approves-novo-nordisks-wegovy-pill-the-first-and-only-oral-glp-1-for-weight-loss-in-adults-302648344.html.


