Type 1 diabetes (T1D) affects far fewer people than type 2, but the stakes for each person are higher. It’s a lifelong, minute-by-minute autoimmune condition. People with T1D depend on insulin to survive, and small improvements in glucose control can translate into fewer emergencies and less organ damage over decades.
T1D accounts for roughly 5-10% of US diabetes diagnoses—about two million individuals—with more than 18,000 youth diagnosed each year. That demographic mix of children, adolescents transitioning to adult care, and adults with decades of exposure helps shape the research questions that matter most.
Advances in diabetes care
Two major shifts are redefining the field of T1D research.
First, connected devices, such as continuous glucose monitors (CGMs) and hybrid closed-loop systems, generate minute-level physiologic signals that can now be linked to downstream outcomes.
Second, immune– and cell-based interventions are beginning to show potential to alter disease course, not just replace insulin.
Together, those advancements create a clear need for real-world datasets that link continuous device data with longitudinal clinical outcomes, allowing researchers to test whether better glucose control today truly reduces hospitalizations, slows kidney decline, or changes long-term risk.
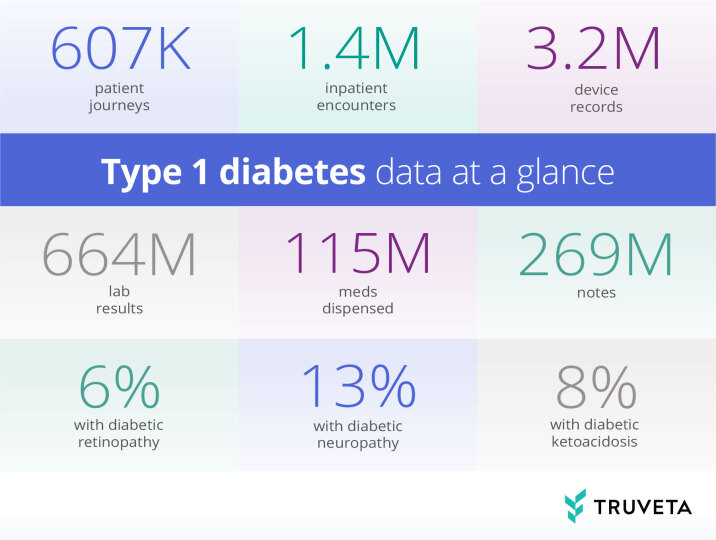
Why use Truveta Data for T1D research
Truveta provides daily updated, complete, representative, and research-ready electronic health record (EHR) data from leading US health systems. Linked with closed claims, Truveta Data offers a longitudinal view of care that supports precise, outcomes-focused research.
For T1D research, this enables:
- Clear visibility into device use, including CGM data captured in routine care (with glucose readings where available) and EHR indicators, such as documented device use.
- Brand-level understanding of devices through unique device identifier (UDI) mapping and a standardized device hierarchy, allowing researchers to study specific CGMs or insulin pumps rather than broad device classes.
- Longitudinal lab trajectories for A1C and creatinine/eGFR, supporting models of glycemic control and kidney function change over time.
- Capture of acute events via emergency department and inpatient encounters to identify patterns of DKA, severe hypoglycemia, and hospital utilization.
- Detailed medication history for insulin regimens and adjunct therapies (including GLP-1s and SGLT2 inhibitors), enabling comparative-effectiveness and safety analyses.
- Linkage to claims and SDOH to support HEOR models, cost analyses, and health equity questions across the full patient journey.
Linking proprietary device data to real-world outcomes with Truveta Live Link
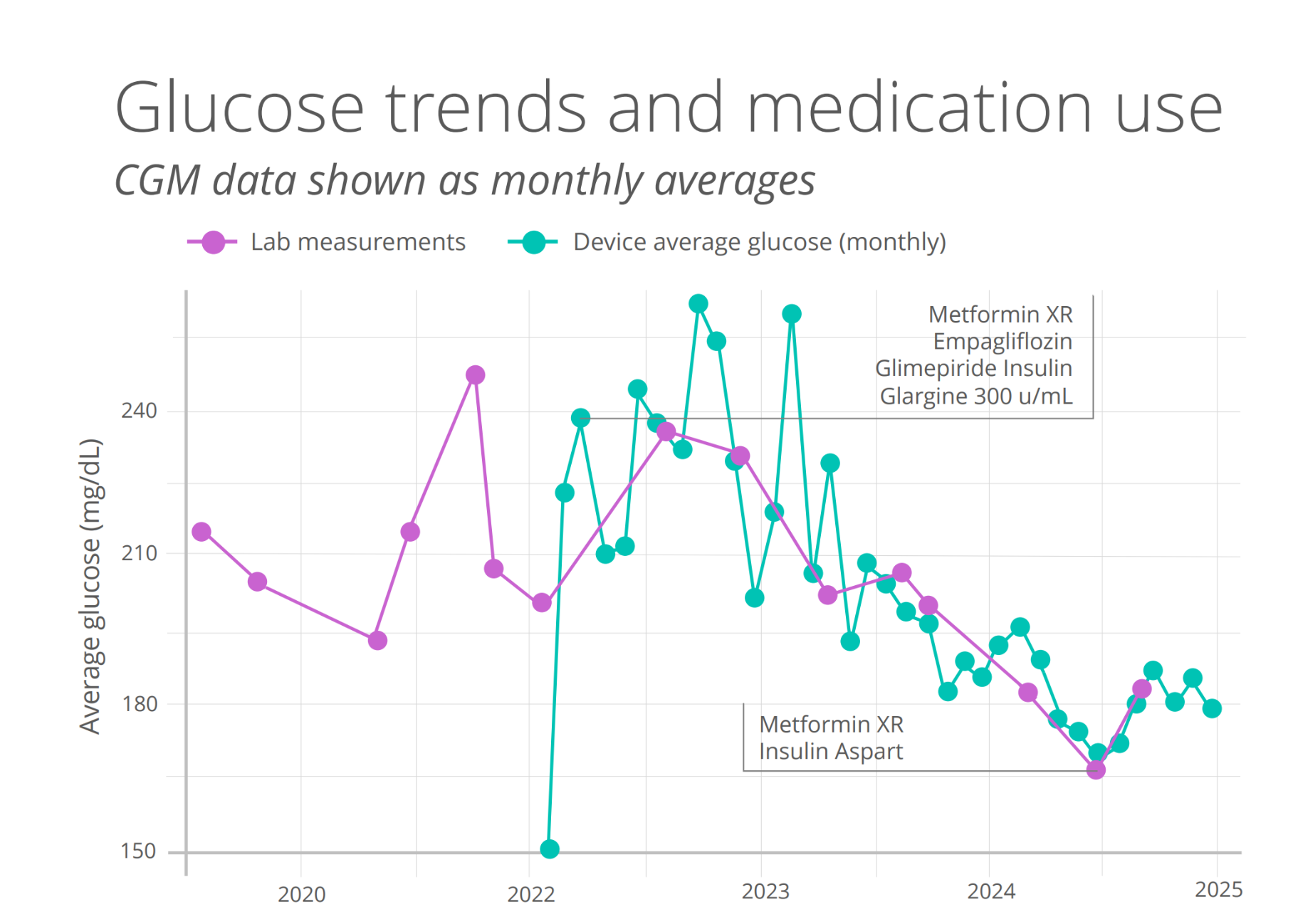
Example of continuous glucose monitoring data linked with longitudinal EHR data, showing monthly average glucose levels alongside medication changes.
As T1D care becomes increasingly driven by connected devices, many of the most important research questions require more than EHR data alone. Researchers need a way to connect proprietary device signals, such as CGM data, with longitudinal clinical and economic outcomes.
Truveta Live Link connects proprietary data with daily updated, complete real-world clinical data—linked to economic data from closed claims—to deliver ongoing evidence across the full patient journey. This approach enables life science organizations to move beyond static, retrospective analyses and generate prospective, regulatory-grade evidence that reflects how therapies and devices perform in routine care over time.
Real-world example: Device signal tied to kidney outcomes
A recent analysis by Dexcom illustrates what becomes possible when propriety CGM data are linked with real-world clinical outcomes at scale.
In an analysis presented in 2025 at the American Diabetes Association and the American Society of Nephrology, adults who initiated continuous glucose monitoring experienced a slower decline in kidney function than similar adults who did not use CGM. The study traces the full evidence chain from device exposure (CGM initiation) to physiologic trajectory (eGFR trend) to clinical outcome (reduced chronic kidney disease progression).
By linking device data with longitudinal clinical context, the analysis demonstrates how continuously connected data can support outcomes-focused research beyond what trials or registries alone can deliver.
Learn more: See how Dexcom uses Truveta Live Link to securely connect proprietary CGM data with real-world clinical outcomes in this case study.
Looking ahead: New biology meets real-world data
Recent research is beginning to hint at how the science of T1D may evolve. A November 2025 Stanford study demonstrated that an immune “reboot” could prevent or reverse the autoimmune disease in mice, offering an early glimpse at therapies aimed at resetting the disease rather than managing glucose levels day to day.
As new innovations move toward human testing, researchers will need datasets that connect genotype, phenotype, device patterns, and long-term clinical outcomes. The Truveta Genome Project is designed for exactly this type of work—building the largest and most diverse linked genomic and clinical dataset to support discovery and accelerate personalized care.

