- Nearly 8% of all prescriptions in March 2026 were for GLP-1 RAs, following the largest quarter-over-quarter percentage point increase in overall GLP-1 RA prescribing observed since the study began in 2019.
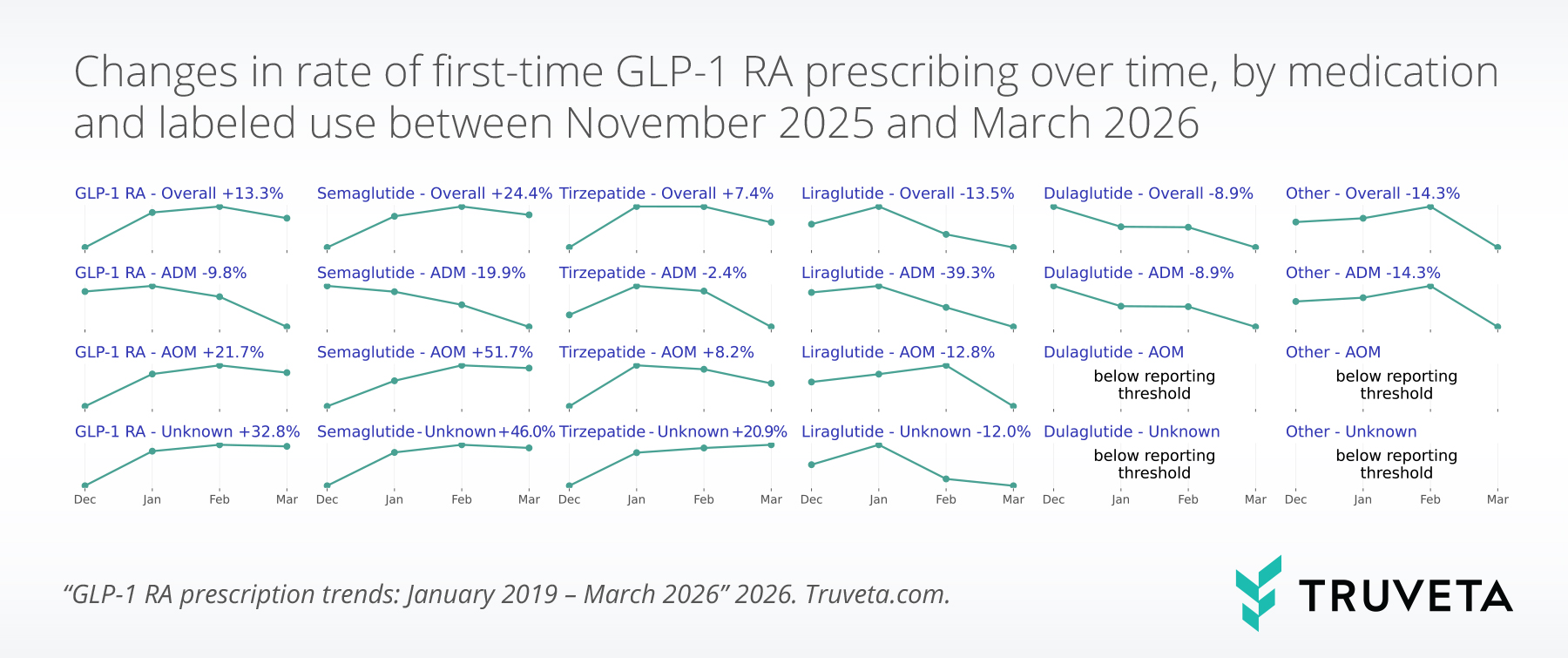
- First-time anti-obesity medication (AOM) prescribing increased 21.7% from December 2025 to March 2026, with first-time AOM semaglutide prescribing increasing by more than 50%.
- AOM semaglutide also showed its largest quarter-over-quarter percentage point increase since the start of the study period, a trend likely driven by approval of the oral formulation of Wegovy in late 2025.
Limited recent data exist on prescribing patterns and patient characteristics for GLP-1 RA medications, whether used as anti-diabetic medication (ADM) for patients with type 2 diabetes (T2D) and/or used as an anti-obesity medication ( for patients with overweight or obesity. Interest in these medications has recently accelerated, although access to and use of GLP-1 RA medications may be impacted by high cost, limited insurance coverage for patients without T2D, and medication shortages.
To offer insight into the latest trends about these medications, Truveta Research has created the GLP-1 RA monitoring report, which will be updated periodically with fresh, timely data. Truveta Data provides the most representative, complete, and timely patient journey data, including full patient medical records, notes, and images, for more than 130 million patients across the US. Truveta Data is also linked with closed claims, mortality, and social drivers of health. Because Truveta Data is updated daily, we can show the latest trends in these medications.
This blog provides a snapshot of the key findings in the most recent report; including prescribing and dispensing medication (indicates whether the patient picked up the medication) trends. For the full analysis—inclusive of demographics, comorbidities, and social drivers of health data for the population, methodology, additional findings, limitations, and citations—you can view the complete report on MedRxiv or directly within Truveta.
Key findings: Prescribing trends
Using a subset of Truveta Data, Truveta Research identified people who were prescribed a GLP-1 RA between January 1, 2019 and March 31, 2026. The report describes prescribing volumes and patient characteristics over time, by medication, and by FDA-labeled use (e.g., ADM, AOM, or unknown).
Overall prescribing trends
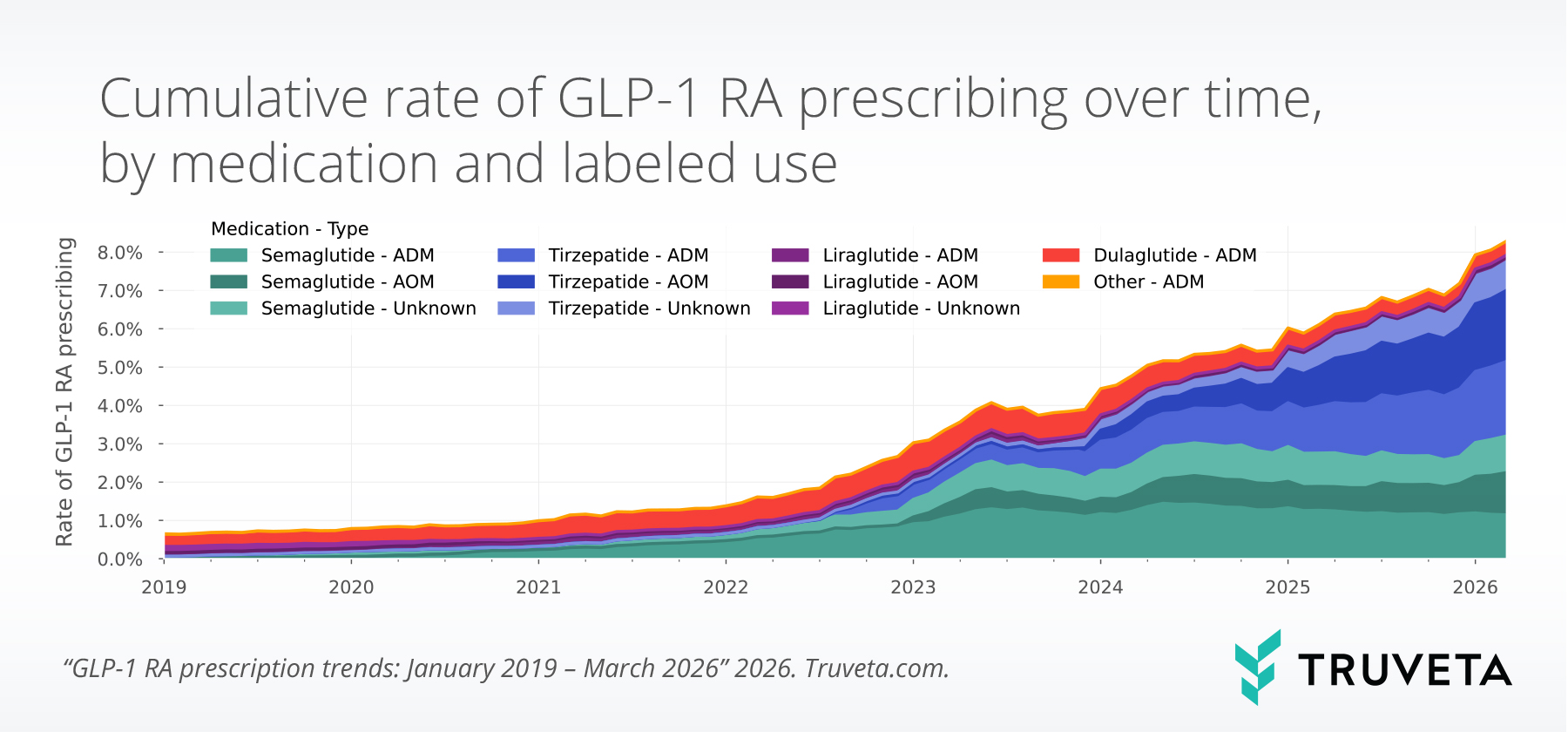
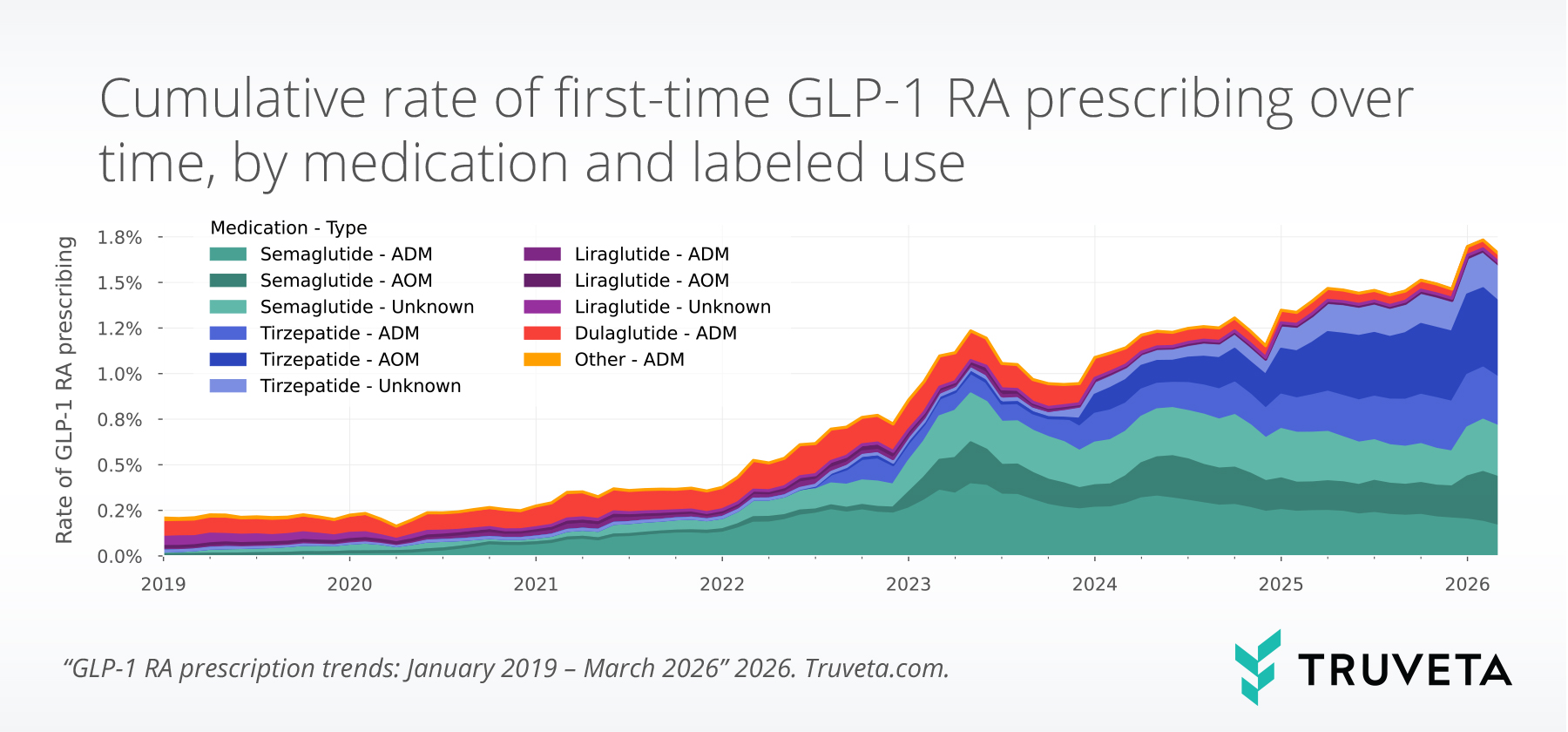
The study found that 2,855,602 patients were prescribed a GLP-1 RA between January 2019 and March 2026, with 14,738,765 total prescriptions during this period.
Overall prescribing rates (GLP-1 RA prescriptions as a proportion of all prescriptions) increased by more than one percentage point from December 2025 to March 2026 (+15.0%), representing the largest quarter-over-quarter percentage point increase observed since the start of the study period in 2019.
As of March 2026, nearly 8 out of every 100 prescriptions were for GLP-1 RAs.
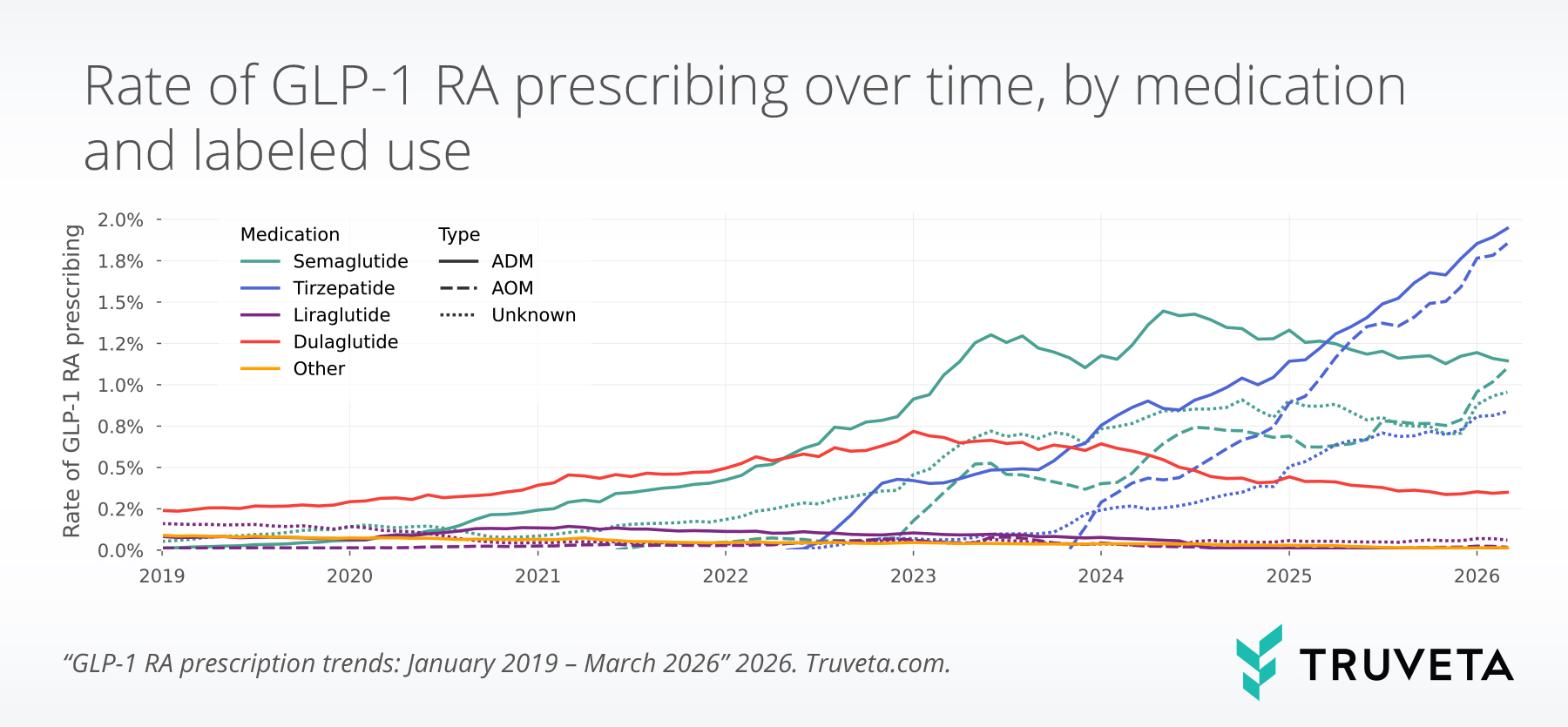
Month-over-month ADM prescribing increased in January, remained stable in February, and increased again in March 2026. Overall, ADM prescribing increased slightly compared to December 2025 (+4.7%).
AOM prescribing increased each month between December 2025 and March 2026, rising from 2.4% to 2.9%, a 23.8% increase overall. The largest increase came from AOM semaglutide, which rose 39.7% over the quarter and posted its largest quarter-over-quarter percentage point increase since 2019.
Tirzepatide remained the most commonly prescribed GLP-1 RA overall, but semaglutide—particularly AOM semaglutide—showed the greatest increase over the quarter.
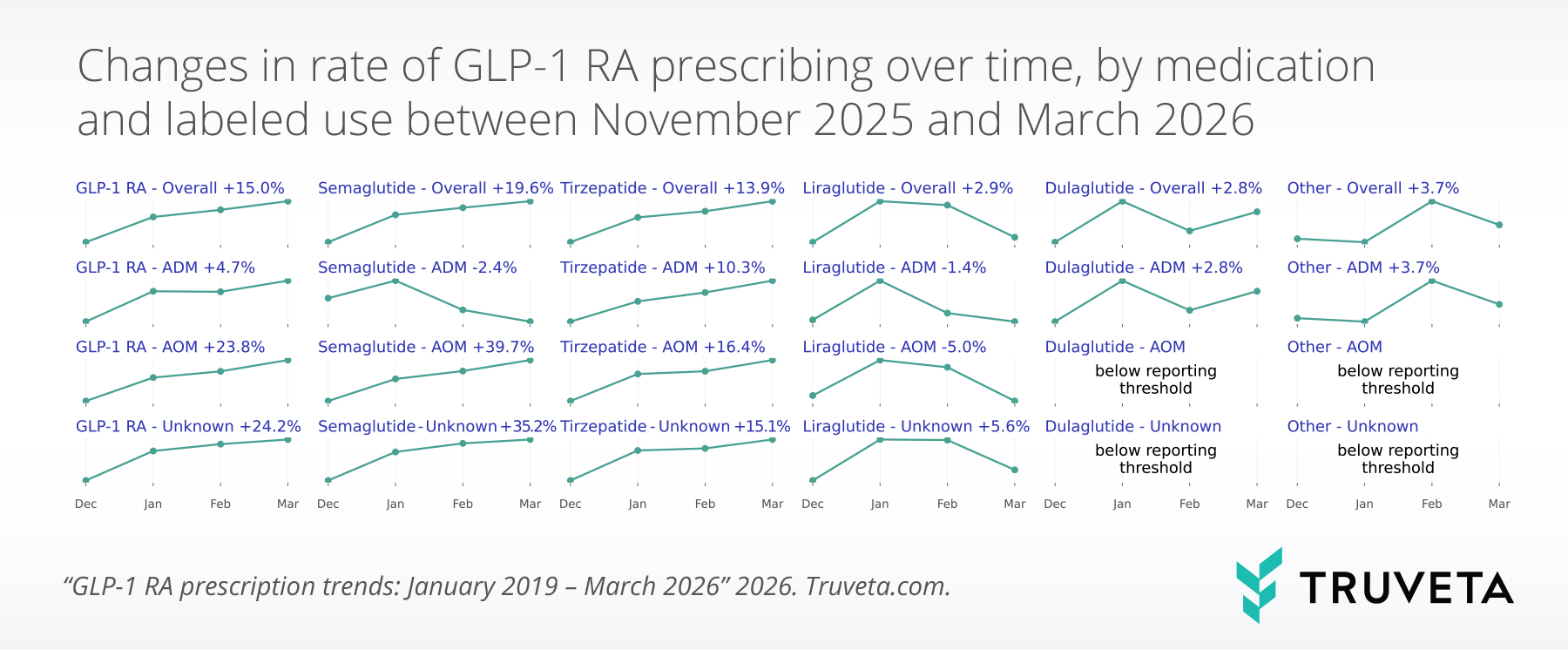
Trends in first-time prescribing
First-time prescribing rates (first-time GLP-1 RA prescriptions per total prescriptions) increased from 1.4% in December 2025 to 1.6% in March 2026 (+13.3%). The increase occurred primarily between December 2025 and January 2026 and then remained elevated through March.
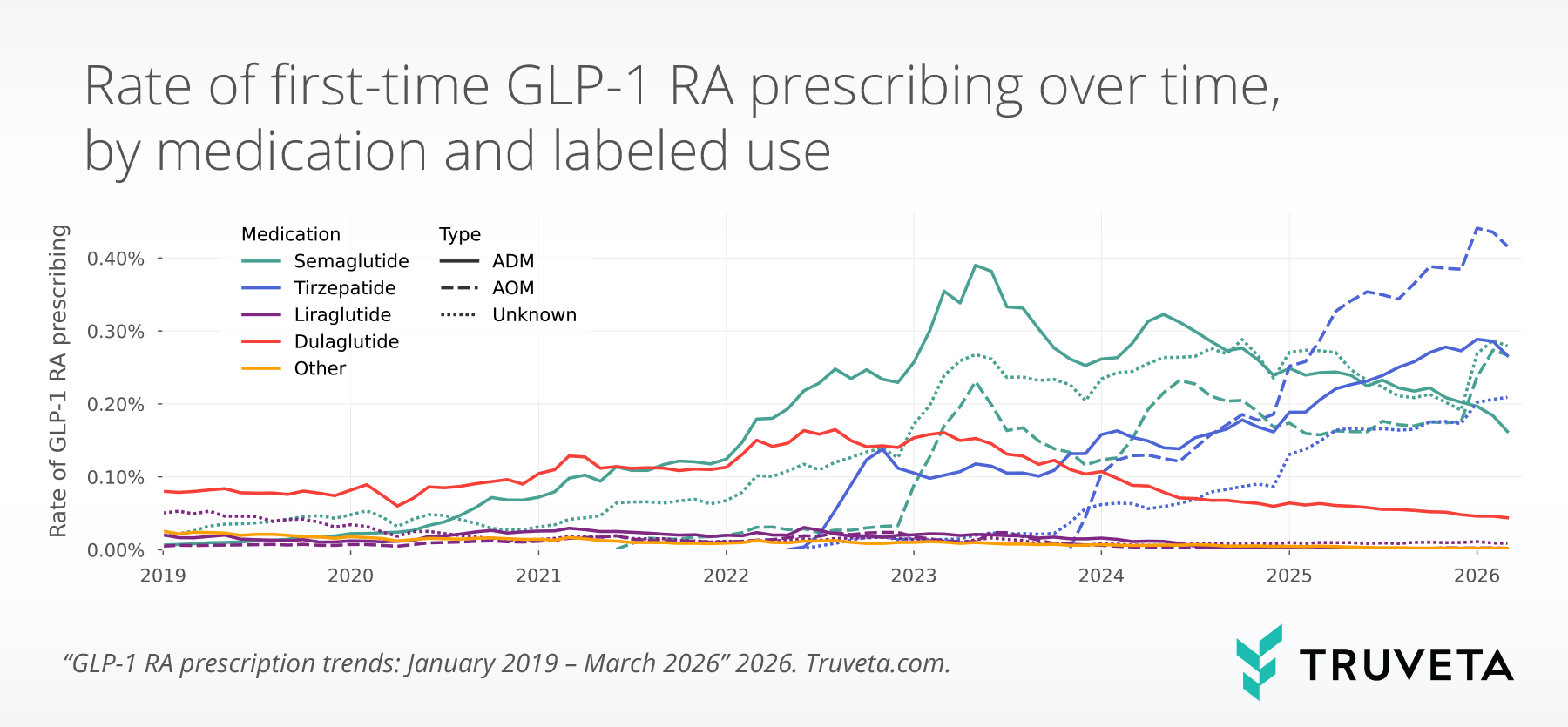
First-time prescribing of ADMs decreased slightly in March 2026 relative to December 2025 (-9.8%).
By contrast, first-time prescribing of AOMs increased 21.7% over the same period, from 0.56% to 0.69%. First-time AOM semaglutide prescribing increased by over 50%, marking the largest quarter-over-quarter increase in first-time AOM semaglutide prescribing since mid-2024, likely driven by the approval of the oral formulation of Wegovy in December 2025.
Discussion
With the popularity of GLP-1 RA medications and challenges in access and insurance coverage, we will continue to monitor prescribing and dispensing trends over time.
The GLP-1 RA monitoring report describes more detailed information about the overall population of patients being prescribed these medications (including demographics and comorbidities), and the proportion and characteristics of patients who filled a GLP-1 RA prescription over time using dispenses. Methodologies, limitations, and citations are also available in the full report. You can also view the full study—including codes, definitions, and more—directly in Truveta.
These are preliminary research findings and not peer reviewed. All data are preliminary and may change as additional data are obtained. These findings are consistent with data accessed April 13, 2026. Data presented in this analysis represent raw counts and/or rates, and post-stratification methods have not been conducted.