In the first comparative effectiveness study of tirzepatide (Mounjaro) and semaglutide (Ozempic) for weight loss using real-world data, Truveta Research found that patients with overweight or obesity taking tirzepatide were three times more likely to achieve 15% weight loss than those taking semaglutide (Ozempic).
Glucagon-like peptide 1 receptor agonist-based (GLP-1 RA) medications, including semaglutide and the dual GLP-1 RA/gastric inhibitory polypeptide (GIP) medication tirzepatide, are used to treat type 2 diabetes or obesity. In this study, Truveta Research explored weight loss trends for patients taking semaglutide and tirzepatide approved by the FDA for treating type 2 diabetes (Ozempic and Mounjaro, respectively).
Unadjusted populations for semaglutide and tirzepatide differ
Using Truveta Data, Truveta Research began with a patient population of over 40,000 patients with overweight or obesity initiating either semaglutide or tirzepatide between May 2022 and September 2023.
The study found that in the initial unadjusted population, those taking tirzepatide and semaglutide differed. While both populations are primarily white and female, those taking tirzepatide are more likely to be younger, have at least some college education, have fewer co-morbidities, and not have type 2 diabetes.
Adjusted population analysis finds tirzepatide more effective than semaglutide for weight loss
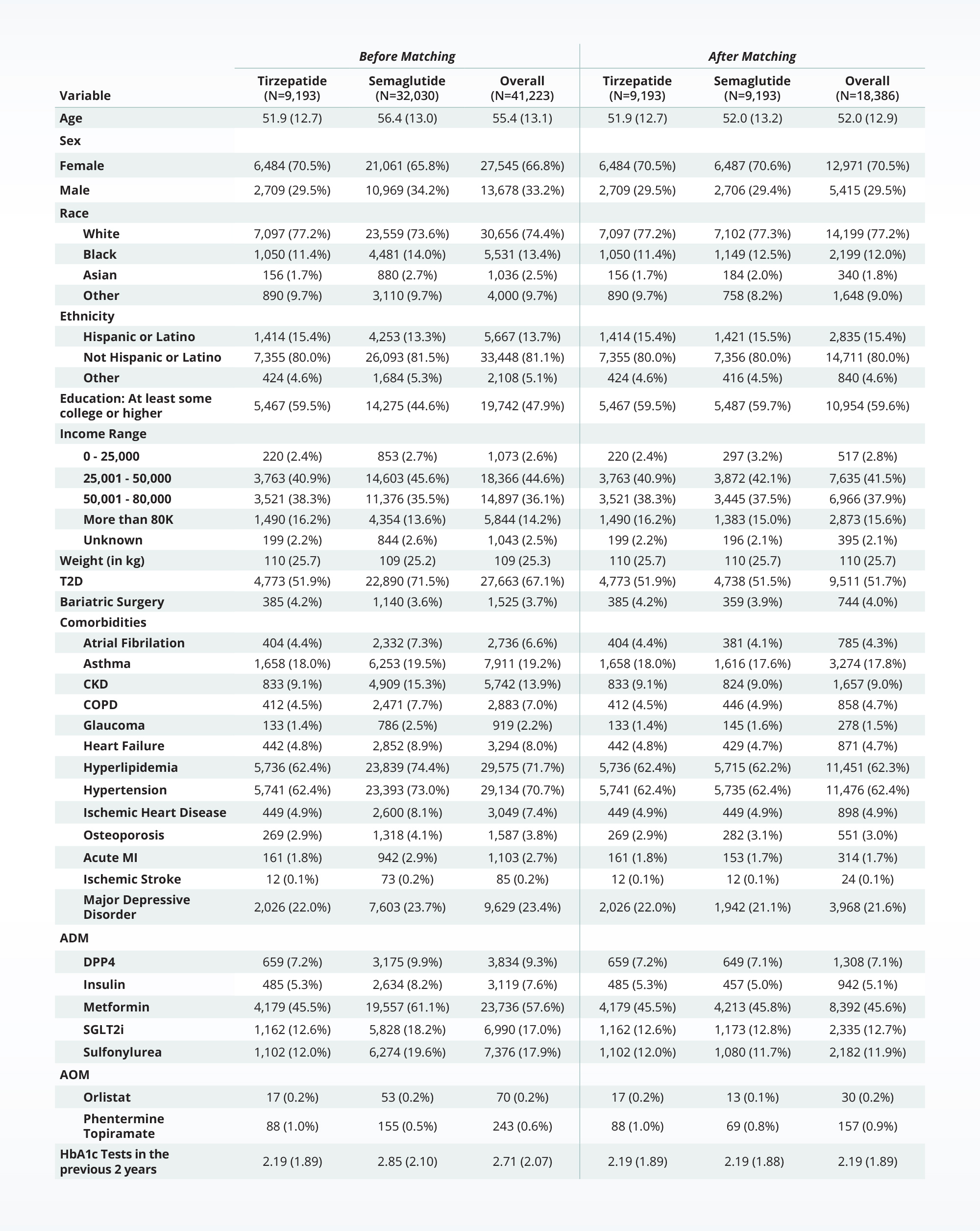
Truveta Research then used multiple rigorous methods (including propensity score matching) to achieve a well-balanced analytic cohort of 18,000 patients to appropriately evaluate the effectiveness of semaglutide compared with tirzepatide for weight loss. The analysis found that patients taking tirzepatide were significantly more likely to achieve weight loss:
- Those taking tirzepatide were 1.8 times more likely than those taking semaglutide to achieve 5% weight loss,
- 2.6 times more likely than those taking semaglutide to achieve 10% weight loss,
- And three times more likely than those taking semaglutide to achieve 15% weight loss.
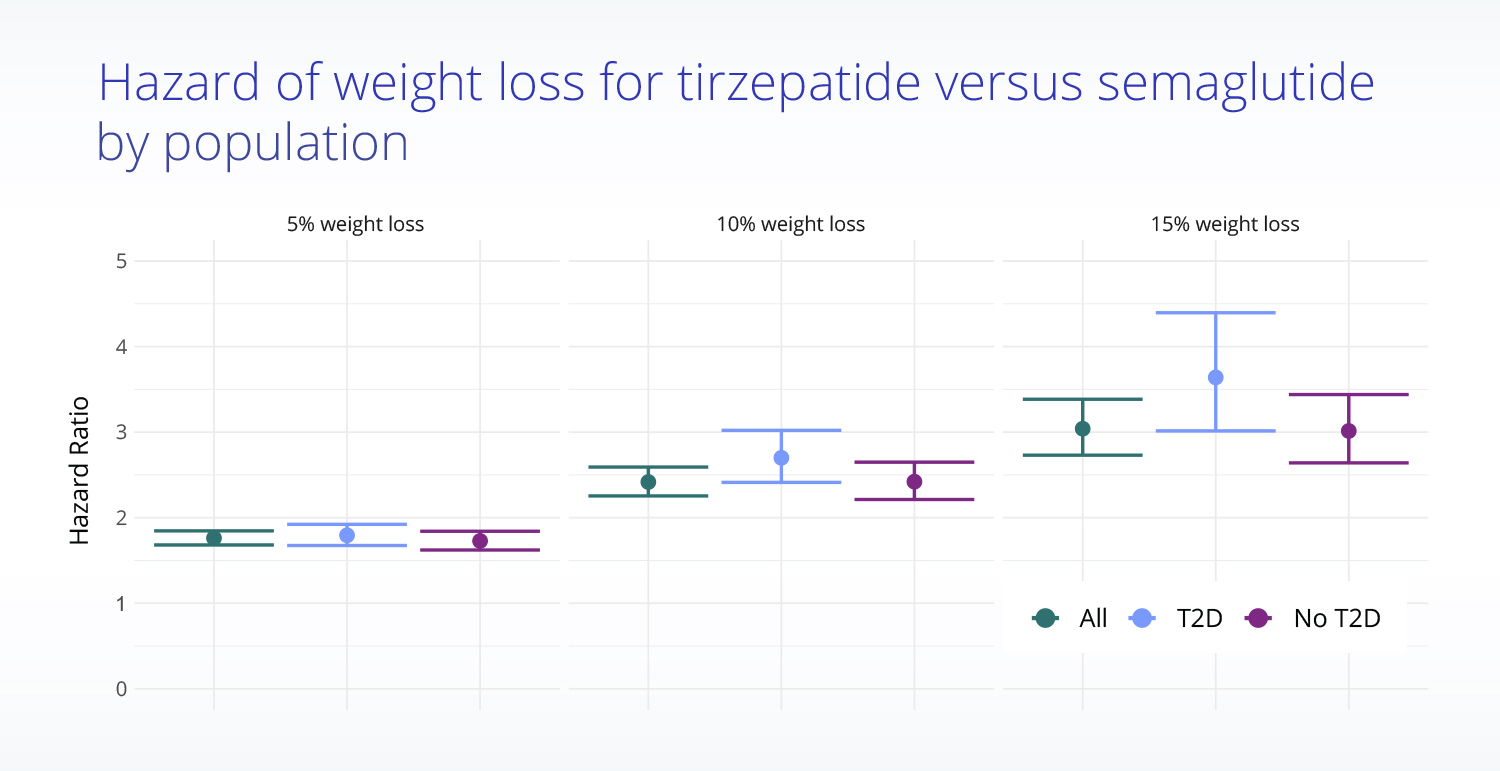
Truveta Research also found that patients taking tirzepatide experienced significantly larger reductions in body weight at specified timepoints:
- At 3 months, the mean percentage change in body weight was -5.9% for those taking tirzepatide versus -3.6% for those taking semaglutide.
- At six months, the mean percentage change in body weight was -10.1% for those taking tirzepatide versus -5.9% for those taking semaglutide.
- At one year, the mean percentage change in body weight was -15.2% for those taking tirzepatide versus -7.9% for those taking semaglutide.
Additionally, Truveta Research found that weight loss was greater for patients without type 2 diabetes than for those with evidence of type 2 diabetes, though differences in effectiveness between tirzepatide and semaglutide were similar.
Rates of gastrointestinal adverse events were similar between those taking tirzepatide and semaglutide.
This research was conducted using Truveta, which provides daily updated, complete EHR data from more than 100 million de-identified patients, including labs, images, medication dispense data, and clinician notes linked with claims and SDOH to accelerate research across all diseases, drugs, and devices. These data were then analyzed using Truveta Studio, which enables scientifically rigorous, fast, and compliant analytics.
Perspective from the study authors
“GLP-1 medications have already changed the way physicians treat type 2 diabetes and obesity, but little real-world data exist to compare the effectiveness of the two most common medications, Ozempic and Mounjaro. While existing trials suggest higher effectiveness of tirzepatide compared to semaglutide for inducing weight loss, head-to-head trials for patients with overweight or obesity are not yet available. It also remains unclear how weight loss observed in these randomized controlled trials will generalize to real-world populations,” said Nick Stucky, MD, PhD, vice president of Truveta Research, practicing infectious diseases physician at Providence Portland Medical Center, and an author on the paper. “That’s what makes today’s study findings so exciting and important. Because Truveta Data provides timely, complete EHR data – including prescriptions and medication dispense data – that captures all patients, not just the insured, we’ve been able to compare the head-to-head efficacy of these two important medications for weight loss in advance of randomized clinical trials. This study can help to inform patient care and outcomes today, not months from now.”
“As a practicing cardiologist and researcher, having the most timely data to inform patient care is paramount,” said Tyler Gluckman, MD, MHA, FACC, FAHA, FASPC, cardiologist at Providence Health, medical director at the Center for Cardiovascular Analytics, Research, and Data Science (CARDS) at Providence Heart Institute, and an author on the paper. “Assessing the real-world effect of semaglutide and tirzepatide on weight loss provides a glimpse into what we may see with the recently approved obesity drug tirzepatide (brand name Zepbound) and how it might compare with semaglutide (brand name Wegovy). Because tirzepatide (brand name Mounjaro) was only approved by the FDA in mid-2022 for type 2 diabetes, the ability to rigorously analyze its use (on- and off-label) for a broad population of patients with overweight or obesity, not just a subset captured in insurance databases, has the power to greatly improve our understanding of how these agents are being used in every day practice and the effect that they’re having.”


